
Articles
These articles are designed to help families make thoughtful, evidence-informed decisions about supplement, skincare, and everyday wellness.
Rather than focusing on trends or product recommendations, each piece explains how to evaluate ingredients, formulation quality, label claims, and risk—so decisions feel clearer and more grounded.
Each article stands on its own wile contributing to a broader framework focused on clarity, safety, and real-world practicality.
What Safety Data Exists for Long-Term Supplement Use in Children?
Many supplements are studied over weeks or months — but what do we actually know about long-term use in children? This article reviews the available safety data, highlights evidence gaps, and explains how to interpret duration and risk over time.
A focused look at the available safety data, and where meaningful gaps still exist.
A supplement often starts with a clear reason: picky eating, a vitamin D concern, a probiotic after antibiotics, a multivitamin during a difficult eating phase. Months later, it may still be part of the daily routine. The original reason made sense, nothing obvious went wrong, and the habit continued.
But tolerating a supplement this week is a different question from knowing whether continued use still makes sense after months or years. Many children take supplements daily for extended periods, yet the evidence base for long-term use is often assumed rather than clearly understood.
This article examines what is known, and what remains uncertain, about the long-term safety of supplement use in children, using an evidence-informed, risk-based approach. Rather than offering reassurances or alarm, it focuses on how safety data is generated, where limitations exist, and how families can think about duration and exposure thoughtfully.
The goal is informed caution, not fear.
Why Long-Term Safety Is a Different Question
Short-term tolerance does not automatically predict long-term appropriateness.¹ A supplement that causes no immediate side effects may still deserve reassessment when used daily, over extended periods, or during critical stages of growth and development. A product can be tolerated and still warrant a second look if the original reason for use has changed.
Long-term exposure introduces questions around accumulation, dose appropriateness over time, and shifting physiological needs.² Understanding this distinction helps explain why long-term pediatric safety data is often more limited than the familiarity of a product might suggest.
Tolerated today does not automatically mean appropriate indefinitely.
Long-term use introduces different questions: cumulative exposure, changing needs, dose over time, and whether the original reason for supplementation still applies.
How This Fits Within an Evidence-First Approach
Evaluating long-term safety requires careful attention to what evidence exists and what does not. The useful question is not "Is this safe forever?" It is "What do we know, what remains uncertain, and does continued use still make sense for this child at this dose?"
This article uses the same evidence-first lens applied throughout this site to examine pediatric supplement safety, with particular focus on duration, exposure, and uncertainty.
Long-term safety is not answered by reassurance alone.
The goal is not to assume harm or safety. It is to understand what evidence exists, what remains uncertain, and how duration, dose, and the child's current needs change the decision.
What "Safety Data" Typically Means in Pediatrics
Families may expect safety data to be one clear answer. In reality, it is often pieced together from different types of evidence.³ In pediatric contexts, safety data typically comes from a mix of sources rather than long-term randomized trials, including short-term clinical studies, observational data, post-market surveillance, and extrapolation from adult data. Each of these contributes information, but none alone provides a complete picture of long-term safety.
Understanding how safety evidence is assembled helps set realistic expectations about what can and cannot be concluded from it.
Sources of Pediatric Supplement Safety Data, and Their Limits
| Evidence Source | What It Can Tell Us | Key Limitations | How to Interpret It |
|---|---|---|---|
| Short-term clinical studies | Tolerance, short-term side effects, early safety signals | Often limited duration, small sample sizes, and narrow populations | Useful for short-term safety, but not enough to assume long-term safety |
| Observational data | Real-world patterns of use and potential associations | Can be affected by confounding factors, inconsistent reporting, and unclear exposure details | Helpful for signals, but rarely definitive on its own |
| Post-market surveillance | Reported adverse events after products are available | Underreporting, incomplete context, and difficulty proving cause | Important for safety signals, but not a full safety profile |
| Adult data extrapolation | General physiology, dose considerations, and known risks in adults | Children are not simply smaller adults; growth and development change the picture | Useful background, but pediatric relevance must be interpreted carefully |
| Upper intake levels and nutrient risk frameworks | Reference points for avoiding excessive intake | Often nutrient-specific and not product-specific | Helpful for dose boundaries, especially with repeated or combined exposure |
| Historical use | Patterns of long-standing use and broad familiarity | Familiar use is not the same as systematic long-term safety evidence | Contextual only; should not replace dose, duration, and risk evaluation |
Long-term pediatric safety data usually comes from multiple evidence sources. Each source can be useful, but none should be treated as a complete answer on its own.
Safety evidence is often pieced together, not handed to families as one clear answer.
That does not mean families should panic. It means long-term decisions deserve more attention to duration, dose, individual need, and periodic reassessment.
Why Long-Term Pediatric Trials Are Rare
There are practical and ethical reasons why long-term supplement trials in children are uncommon.⁴ Challenges include ethical constraints around prolonged exposure, difficulty controlling diet and environment over time, limited funding for non-pharmaceutical interventions, and rapidly changing nutritional needs during growth.
This does not mean no one cares about long-term safety. It means these studies are genuinely difficult to conduct well. The absence of long-term data often reflects structural limitations in how research is resourced and designed, not negligence.
A lack of long-term trials does not automatically mean danger, but it does limit certainty.
For families, that uncertainty should lead to proportionate caution: define the reason for use, avoid unnecessary exposure, and revisit the decision as needs change.
What Can Be Inferred, and What Cannot
In some cases, long-term safety is inferred from known nutrient physiology, established upper intake levels, and patterns of historical use. These inferences can be useful, but they are not equivalent to direct evidence.⁵ Useful inference is not the same as a complete answer.
Evidence is typically tied to specific compounds, doses, and contexts, not to supplement categories as a whole. Understanding this distinction helps prevent overconfidence in generalized safety claims.
What Can Be Inferred, and What Still Needs Caution
Dose, Duration, and Accumulation Matter
Long-term safety is rarely about whether a nutrient is "safe" in isolation. The question changes when a supplement becomes part of the daily background. Cumulative exposure, dose relative to need, duration of use, and changes in diet over time all shape how appropriate continued use remains.
Form and dose tend to matter more than ingredient count alone, especially with ongoing use.⁶ A dose appropriate for short-term support may not remain appropriate indefinitely, particularly as a child's diet, weight, and nutritional needs evolve.
Children's Needs Change Over Time
Children do not stay in the same nutritional situation for long.⁷ Growth, development, and dietary patterns shift continuously. A supplement that made sense during a season of picky eating, low intake, or recovery may not need to remain in the routine indefinitely. What was reasonable at one stage may become unnecessary, or even excessive, later.
This is why long-term supplement decisions benefit from periodic reassessment rather than automatic continuation.
For ongoing supplement use, reassess periodically:
Common Assumptions About Long-Term Use
Several assumptions often appear in discussions of long-term supplementation: "It's just a vitamin, so it's harmless." "More coverage is better than less." "If it hasn't caused problems yet, it won't."
These assumptions are understandable. Supplements often feel familiar and low-risk, and that familiarity can make continued use feel like a neutral choice. But familiar does not mean there is nothing to evaluate. These assumptions can quietly obscure more relevant questions about necessity, dose, and duration.⁸
Familiar does not automatically mean risk-free.
The most relevant safety questions are not whether a supplement feels common or gentle. They are whether the dose, duration, and ongoing need still make sense.
Risk Management, Not Zero Risk
No intervention, dietary or otherwise, is entirely risk-free. The goal is not to eliminate every possible risk. It is to make the decision proportional to the need. Long-term safety decisions involve weighing potential benefit, potential risk, likelihood of deficiency, and the availability of alternative approaches.⁹
This framing aligns with how safety decisions are made in other areas of child health, where the aim is proportional, thoughtful evaluation rather than certainty.
A Practical Way to Think About Duration
Rather than viewing supplementation as an open-ended decision, it helps to ask: What prompted supplementation initially? Has that reason changed? Is continued use still justified? Should dose or form be revisited?
Duration should be part of the decision, not something that disappears once the supplement becomes routine. A time-bounded approach supports more intentional use and makes it easier to adjust as the child's needs evolve.
Long-term supplementation should be revisited, not put on autopilot.
A supplement that made sense at one point may not need to continue indefinitely. A time-bounded review helps families make changes without guilt, fear, or unnecessary complexity.
Using a Structured Decision Tool
Long-term supplementation decisions benefit from clarity around purpose, duration, and reassessment. A structured decision flow can help think through need, exposure, and proportional risk, especially when use extends beyond short-term support.
Use the decision flow to review need, exposure, duration, and proportional risk when supplement use extends beyond short-term support.
Use the Supplement Decision Flow →Use the checklist to review ingredient identity, dose, formulation quality, and evidence signals before relying on supplement claims or continuing routine use.
View the Checklist →Why This Matters for Families
Families often continue supplements because the original reason made sense, the product is already in the routine, and nothing obvious has gone wrong. That is understandable. Starting a supplement for a defined reason and tolerating it without issue does not mean the original reason is still present.
Children's diets, growth patterns, and needs change. Revisiting long-term supplement use helps families make adjustments without fear, guilt, or the feeling that they did something wrong. The question is not whether starting the supplement was a mistake. The question is whether continuing it still fits the current situation.
Explore structured EFW resources for evaluating supplement tradeoffs, including dose, duration, exposure, formulation quality, and real-world use.
Explore the Frameworks Hub →Pulling It All Together
Long-term supplement use in children is an area where certainty is often assumed but evidence is more nuanced. An evidence-informed approach acknowledges those limits, prioritizes dose and duration, and treats supplementation as a revisitable decision, not a permanent one.
A supplement is easiest to keep when it becomes part of the routine. That is exactly why long-term use deserves a periodic pause. The most useful question is not "Is this supplement safe forever?" It is whether continued use meaningfully reduces risk in this child, at this stage, and at this dose.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- Ioannidis JPA. Why Most Clinical Research Is Not Useful. PLOS Medicine.
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes: Tolerable Upper Intake Levels.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know About Safety.
- U.S. Department of Health & Human Services. Ethical Considerations in Pediatric Research.
- National Institutes of Health, Office of Dietary Supplements. Dietary Supplement Fact Sheets (Safety and Physiology Sections).
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes: Risk Assessment Framework.
- Centers for Disease Control and Prevention. Nutrition, Growth, and Development Guidance.
- National Institutes of Health, Office of Dietary Supplements. Fat-Soluble Vitamins Fact Sheets (Vitamin A, D, E, K).
- National Academies of Sciences, Engineering, and Medicine. Framework for Assessing Nutrient Risk and Benefit.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Do Children Actually Need Supplements? A Risk-Based Perspective
An evidence-informed look at whether healthy children need supplements, examining common assumptions, nutritional adequacy, and when supplementation may—or may not—be warranted.
A risk-based approach to evaluating when supplementation may be appropriate, and when it may not be necessary.
A child goes through a picky eating phase and you wonder whether a multivitamin is a reasonable precaution. A label suggests the product supports growth, immunity, or healthy development. A friend uses one; a pediatrician says diet is fine. You are trying to decide whether doing nothing is enough, or whether adding something is the responsible choice.
The question of whether children need supplements is often framed in absolute terms. Some messaging suggests supplements are essential for healthy development, while other guidance implies they are unnecessary or even excessive. For parents trying to make thoughtful decisions, this all-or-nothing framing rarely helps.
This article approaches the question from a risk-based, evidence-informed perspective. Rather than asking whether children should take supplements, it explores when supplementation may be reasonable, when it is unlikely to add value, and how to think about risk, adequacy, and context.
The goal is not to promote supplementation, but to replace confusion with clarity.
Why This Question Is So Hard to Answer
Part of what makes this question difficult is that both extremes can feel convincing. One side makes supplements sound necessary for healthy development. The other makes any supplement for a child feel excessive or unnecessary. Most real family decisions sit somewhere in between.
Children are not a single population. Nutrient needs vary by age, growth stage, diet, health status, and environment. As a result, blanket statements about supplementation often fail to reflect real-world variability.
At the same time, supplement marketing tends to focus on reassurance, promising to fill gaps or support immunity without clearly defining what those gaps are or how often they exist in a given child's diet. Understanding whether supplements are needed requires stepping back from product categories and focusing on risk and context.
How This Fits Within an Evidence-First Approach
Across this site, supplements are evaluated as conditional tools, not defaults. The useful question is not whether a supplement sounds helpful. It is whether it addresses a defined concern in a way that is appropriate for the child, the dose, and the duration.
This article uses that same evidence-first lens to evaluate pediatric supplementation by considering dietary adequacy, risk factors, and potential tradeoffs, rather than assuming supplements are either necessary or unnecessary. For a broader look at how to apply this thinking to a specific supplement decision, see How to Decide Whether a Supplement Is Worth Trying at All.
What Pediatric Guidance Actually Emphasizes
Mainstream pediatric guidance generally does not recommend routine supplementation for all children.¹ Instead, it emphasizes meeting nutrient needs primarily through food,² identifying specific risk factors for deficiency, and using targeted supplementation when appropriate.³
This does not mean supplements are never useful. It means the reason for use should be clear. Supplements are situational, not universal.⁴ Understanding this helps parents avoid both over-supplementation and unnecessary worry about whether their child is missing something.
Dietary Adequacy Comes First
Before considering supplements, it helps to assess whether a child's diet is likely meeting basic nutritional needs. A child does not need to eat perfectly for food to contribute meaningfully to nutrient needs. A reasonably varied diet, regular intake of fortified staple foods, and consistent growth and development can all support adequacy. In these situations, routine supplementation may offer limited benefit.
That said, adequacy is not always easy to assess from the outside, and dietary patterns vary widely between families. Picky eating, food preferences, and inconsistent intake are all common and do not automatically signal a deficiency. How to Read a Supplement Label (Without Getting Misled) can help clarify what label information actually tells you, and what it does not.
When Supplementation May Be Reasonable
Supplementation is more reasonable when there is a clear reason for use, not just a general desire to cover everything. Situations where targeted supplementation is more commonly considered include picky or restrictive eating patterns, medically indicated dietary limitations, periods of rapid growth, limited sun exposure or geographic factors, and specific life stages or transitions.
In these cases, supplements are typically used to address a defined concern, not as a blanket safeguard. This purpose-first approach mirrors how supplements are evaluated throughout this site.
Risk Factors Matter More Than Averages
Population-level recommendations describe averages. Individual children do not live at the average.⁵ Risk-based evaluation asks different questions: Is there a realistic reason this child might not be meeting needs through diet alone? Are there signs suggesting a potential gap? Would supplementation meaningfully reduce risk, or simply add complexity?
This approach avoids both unnecessary supplementation and rigid adherence to generalized guidance that may not fit the child in front of you.
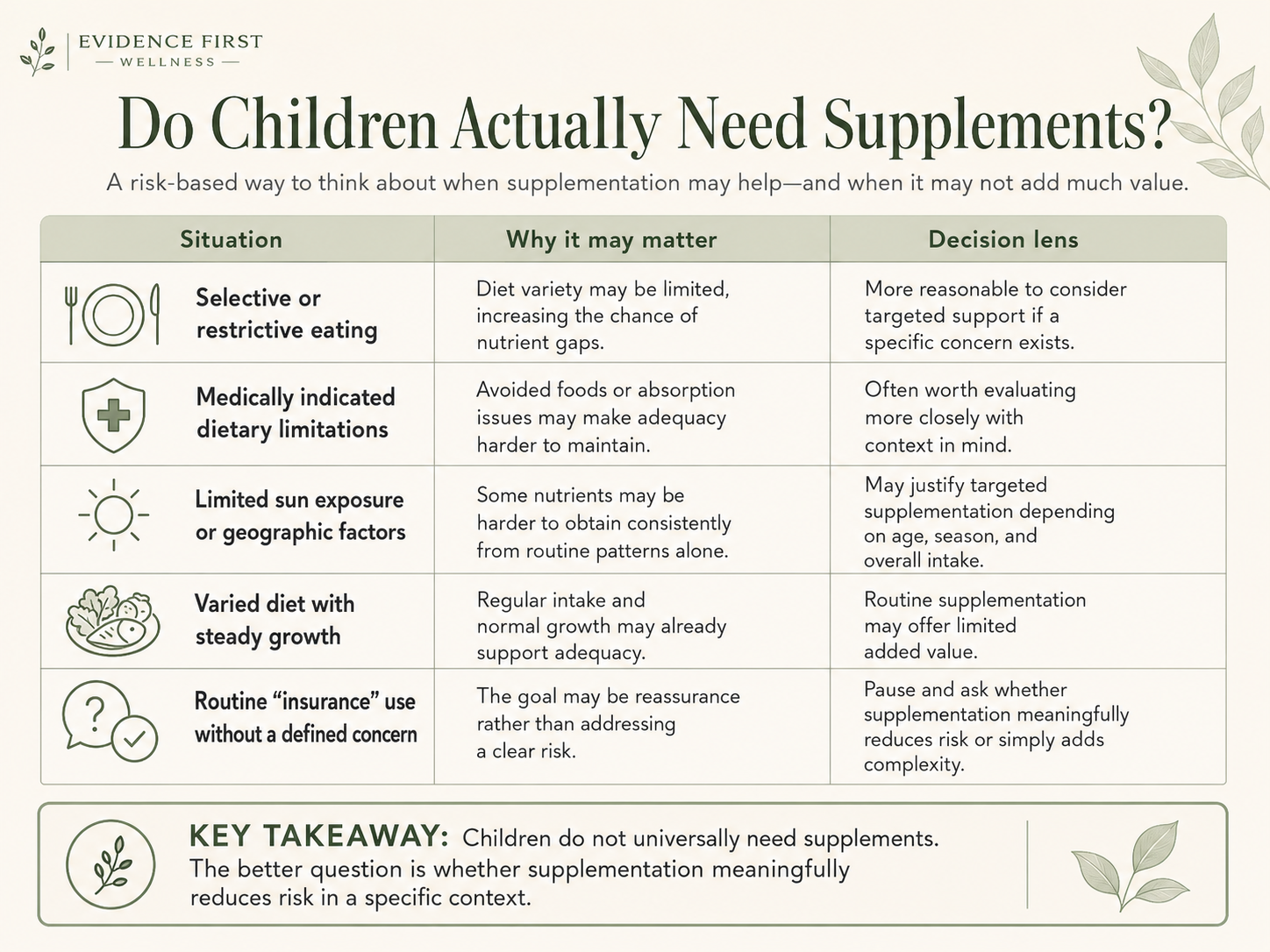
When Supplementation May Be Reasonable vs. When It May Offer Limited Value
| Situation | Why It May Matter | Decision Lens |
|---|---|---|
| Picky or restrictive eating | Limited dietary variety may reduce intake of certain nutrients | Define the specific gap and match the supplement to it |
| Medically indicated dietary limitations | Exclusion of food groups may affect nutrient adequacy | Use targeted supplementation in line with professional guidance |
| Limited sun exposure or geographic factors | Vitamin D synthesis depends on sunlight exposure, which varies significantly | Situational supplementation may be reasonable; dose and form still matter |
| Varied diet with steady growth | Dietary adequacy may already be met through food | Routine supplementation may add limited benefit; evaluate before adding |
| Routine "insurance" use without a defined concern | Motivation is reassurance, not a specific risk | Clarify what is being addressed before continuing; reassess duration |
This table is not a recommendation to supplement or not to supplement. It is a way to match context to decision-making rather than defaulting to either extreme.
Potential Downsides of Routine Supplement Use
While many supplements are well tolerated, routine use without a clear rationale can introduce tradeoffs. These may include unnecessary nutrient exposure, dosing that exceeds needs over time,⁶ reliance on supplements instead of dietary variety, and added ingredients that are not well studied in children.
Recognizing these tradeoffs helps keep decisions proportional rather than fear-driven. For a focused look at long-term use specifically, What Safety Data Exists for Long-Term Supplement Use in Children? examines where the evidence is strong and where meaningful gaps remain.
Dose, Form, and Duration Still Matter
Even when supplementation is considered reasonable, how it is used matters. A supplement can be a reasonable choice in concept and still be poorly matched if the dose, form, or duration does not fit the situation. Form and dose tend to matter more than ingredient count alone, particularly in pediatric contexts.
Duration also matters. Short-term, targeted supplementation differs meaningfully from long-term routine use. The Frameworks & Decision Tools Hub includes structured tools for thinking through these variables across different supplement categories.
Children Are Not Small Adults
One of the most common pitfalls in supplement decision-making is extending adult practices to children.⁷ Children differ from adults in metabolism, body size and composition, tolerance thresholds, and the available evidence base. Adult research does not automatically apply to children, and dose assumptions in particular require careful calibration to age and context.
This is one reason adult supplement habits should not automatically become children's routines. What is appropriate or well-studied in adults may not translate directly, and the gap in pediatric-specific evidence is worth keeping in mind.
Managing Expectations Around "Insurance" Supplements
The instinct to want reassurance is reasonable. When a child's diet is uncertain or inconsistent, adding a supplement can feel like a responsible precaution. The issue is whether the product actually answers the concern.
Supplements used as nutritional "insurance" can obscure important questions: Insurance against what, exactly? For how long? At what dose? Without clear answers, supplementation can drift from a temporary support into an unexamined routine that no longer reflects the original reason for starting it.
A Practical Way to Think About the Decision
Rather than asking whether children need supplements, a more useful approach is to ask: Is there a specific concern being addressed? Is supplementation likely to meaningfully reduce risk? Are form and dose appropriate for age and use? Is this decision intended to be temporary or ongoing?
This reframing keeps the focus on meaningful risk reduction, not adding supplements by default.⁸
The Evidence-Informed Family Supplement Guide is coming soon. It will walk through children's multivitamins, probiotics, magnesium, label claims, dosing, safety tradeoffs, and reusable worksheets for evaluating family supplement decisions with more clarity.
See What's Coming →A Structured Way to Walk Through the Decision
If you are unsure whether supplementation is appropriate in your situation, the Supplement Decision Flow provides a step-by-step way to think through dietary adequacy, risk factors, dose considerations, and duration. It is not a recommendation tool. It is a structured pause.
Use the decision flow to work through dietary adequacy, risk factors, dose considerations, and duration before committing to supplementation. No brand rankings. No product recommendations.
Use the Supplement Decision Flow →Why This Matters for Families
Parents are often making these decisions in the middle of real life: picky eating, busy routines, growth questions, school germs, advice from pediatricians, family members, and product labels that can make nutrient gaps feel more significant than they may be.
A risk-based approach helps families avoid both extremes: adding supplements out of general concern, and dismissing reasonable targeted support when a defined need exists. The goal is not to arrive at a fixed answer about whether supplements are worthwhile. It is to arrive at the right answer for this child, in this situation, for a realistic duration.
Pulling It All Together
Children do not universally need supplements, but some children may benefit from targeted supplementation in specific contexts. An evidence-informed approach avoids extremes. It prioritizes dietary adequacy, evaluates risk thoughtfully, and uses supplements as tools rather than defaults.
A supplement is most useful when it addresses a defined concern, at an appropriate dose, for a realistic duration, with tradeoffs that make sense for the child and family. The most useful question is not "Should children take supplements?" but whether supplementation meaningfully reduces risk in this situation.
Use the checklist to review ingredient identity, dosing, formulation quality, and supplement claims before deciding whether a product fits the need.
View the Checklist →If you're weighing a specific supplement for your child, you can submit a question to help guide future Evidence First Wellness content. This isn't medical advice, but reader questions help shape future explanations about evidence, formulation, and common marketing claims.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- American Academy of Pediatrics. Where We Stand: Vitamin Supplements for Children.
- National Institutes of Health, Office of Dietary Supplements. Multivitamin/Mineral Supplements Fact Sheet.
- Centers for Disease Control and Prevention. Nutrition for Children and Adolescents.
- Harvard T.H. Chan School of Public Health. The Nutrition Source: Do Children Need Vitamin Supplements?
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes (Population-Based Recommendations).
- National Academies of Sciences, Engineering, and Medicine. Tolerable Upper Intake Levels for Vitamins and Minerals.
- National Institutes of Health. Pediatric Research and Age-Specific Dosing Considerations.
- National Academies of Sciences, Engineering, and Medicine. Risk Assessment Framework for Nutrient Intake.
All sources are freely accessible via NIH, FDA, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
How to Interpret Supplement Research Without Getting Misled
A practical, evidence-informed guide to interpreting supplement research, explaining common study limitations, how findings are often overstated, and how to read claims with more clarity and context.
A practical guide to understanding supplement research and distinguishing strong evidence from weak signals.
A product label says "clinically studied." A product page cites a study but does not explain who was studied or at what dose. A post online shares a study as proof that a supplement works. You are trying to decide whether any of it actually applies to your child or family situation. The research language sounds reassuring, but it does not always give you enough information to know whether it should.
Supplement research is frequently referenced in marketing materials, product descriptions, and online discussions, but rarely explained in a way that helps families understand what the findings actually mean.
For families trying to make thoughtful decisions, the challenge is not finding research. It is learning how to interpret it without extending conclusions beyond what the data supports.
This article explains how to approach supplement research using an evidence-informed approach. Rather than encouraging skepticism for its own sake, it focuses on understanding scope, limitations, and common sources of misinterpretation.
The goal is clarity, not cynicism.
Why Supplement Research Is So Easy to Misinterpret
Most supplement studies are conducted under specific and controlled conditions: defined populations, fixed doses, limited timeframes, and narrowly measured outcomes.¹ When those findings are translated into marketing language, much of that context disappears.
The problem is not always the study itself. Often, the problem is what happens when a narrow finding is translated into a broad claim. A study conducted in healthy adults may be used to justify claims for children, or results tied to a single compound may be generalized to an entire product category.² The finding can be valid in one context and still be less relevant in another.
Understanding that translation gap matters more than memorizing research methodology.
How This Fits Within an Evidence-First Approach
Interpreting research responsibly requires separating what a study actually shows from what is implied or assumed. The useful question is not simply "Is there a study?" It is "Does this study actually support the claim being made?"
This article uses that same evidence-first lens to evaluate research claims carefully, without dismissing evidence or overstating its implications.
How evidence is selected, interpreted, and cited across this site is explained in How We Cite Evidence.
The steps below walk through supplement research systematically, focusing on interpretation rather than conclusions.

Step One: Identify What Was Actually Studied
Before interpreting a claim, it helps to clarify who was studied (age, health status, baseline nutrition), the specific compound or form tested, the dose and duration, and the outcome measured.
A study in adults does not automatically answer a question about children. A study on one form of an ingredient does not automatically apply to a different form or to a multi-ingredient product. Evidence is typically tied to specific compounds and contexts, not broad supplement categories.³
Step Two: Distinguish Outcomes From Implications
Many supplement studies measure intermediate outcomes such as biomarker changes or short-term physiological shifts.⁴ These findings can be informative, but a measurable change is not automatically the same as a meaningful benefit.
When a claim moves from "this marker changed" to "this supplement improves health," interpretation has already begun to stretch. Understanding what was measured and what was not helps keep conclusions proportional to the data.
Step Three: Consider Form, Dose, and Duration
Research findings are closely tied to how an ingredient was delivered and used. If the study used a different form, a different dose, or a different duration than the product being considered, the relevance of that evidence changes. Differences in chemical form, dose level, frequency, and study duration⁵ can meaningfully shift outcomes.
Ingredient name alone is not enough to assume the research applies. This is explored in more depth in Why Dosage and Form Matter More Than Ingredient Lists.
Step Four: Recognize the Limits of Individual Studies
Single studies rarely provide definitive answers. Most evidence develops gradually through replication, consistency across different populations, and systematic reviews.⁶ One study can be useful, but it should not carry more weight than it can support.
This is especially relevant in supplement research, where small or narrowly scoped studies, study design choices, funding sources, and publication bias can all influence how results are framed and presented.
Common Types of Supplement Studies, and What They Can and Cannot Tell Us
Not all research carries the same weight. Study type matters when interpreting supplement claims.⁷
In vitro studies are conducted in cells or test tubes. These studies help explore biological mechanisms but do not show how a supplement behaves in the human body.
Animal studies are useful for generating hypotheses and understanding potential pathways, but results may not translate directly to humans.
Small human trials are often short-term and narrowly defined. These studies can suggest potential effects under controlled conditions but may not reflect real-world use or broader populations.
Observational studies identify associations rather than causation. These findings are especially prone to overinterpretation.
Each study type contributes differently to the evidence base. Understanding these distinctions helps prevent treating early or limited findings as definitive conclusions.
Step Five: Separate Evidence from Plausibility
Some supplement claims are framed as "biologically plausible," meaning they align with known mechanisms. While plausibility can guide research, it is not the same as demonstrated benefit.⁸ A mechanism can make a claim worth studying. It does not make the claim proven.
Treating plausibility as proof is one of the most common ways hypotheses get mistaken for conclusions.
Common Ways Research Is Used in Marketing
Research language can be technically accurate and still be incomplete for decision-making. Common patterns in supplement marketing include citing studies without specifying the population or dose, using ingredient-level research to support claims about multi-ingredient products, and implying long-term benefits based on short-term findings.
Each of these patterns can make a claim sound more settled than it is. Recognizing them helps families engage with research more critically without dismissing it entirely. For a closer look at one of the most common examples, see What "Clinically Studied" Really Means on a Supplement Label.
A Practical Test for Research Claims
When encountering a research-based claim, it can help to pause and ask a few direct questions. Who was studied? What form and dose were used? Is the outcome measured meaningful, or indirect? Is the conclusion proportional to the data, or extended beyond it?
If the claim does not give enough information to answer those questions, it may be more persuasive than useful. For a more structured way to work through these questions, the Frameworks & Decision Tools Hub and Supplement Decision Tool are designed to support exactly this kind of evaluation.
The Evidence-Informed Family Supplement Guide is coming soon. It will turn research interpretation, label review, and category-specific supplement questions into reusable worksheets and decision prompts for families.
See What's Coming →Use the checklist to review ingredient identity, dosing, formulation quality, and what supplement research claims actually mean before relying on them.
View the Checklist →Why This Matters for Families
Parents rarely have time to read a full study. Research language on a product page can make something feel more credible than the underlying evidence warrants, because the missing context is not visible. A single study can sound convincing when the details about population, dose, form, and duration are not part of the claim.
Better interpretation does not require expertise. It requires knowing which questions to ask. Understanding how to read research without overextending its conclusions helps reduce anxiety, avoid unnecessary supplementation, and support more deliberate decisions. For broader context on when supplementation may be worth considering, see How to Decide Whether a Supplement Is Worth Trying at All.
Pulling It All Together
Interpreting supplement research does not require advanced training, but it does require attention to context. The most useful questions when encountering a research claim are: Who was studied? What exactly was tested? At what dose and for how long? What outcome was measured? What assumptions are being made beyond the data?
Research should help narrow a question, not close it before the context is understood. Answering these questions keeps research in its proper role: informative, but not definitive on its own.
If you are also thinking about how labels present this kind of information, How to Read a Supplement Label (Without Getting Misled) applies the same interpretive lens to what appears on the front and back of a product.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- National Institutes of Health. Understanding Clinical Research.
- Ioannidis JPA. Why Most Published Research Findings Are False. PLOS Medicine.
- U.S. Food & Drug Administration. Dietary Supplement Labeling Guide.
- Fleming TR, DeMets DL. Surrogate End Points in Clinical Trials. New England Journal of Medicine.
- Heaney RP. Factors Influencing the Measurement of Bioavailability. American Journal of Clinical Nutrition.
- Ioannidis JPA. Why Most Clinical Research Is Not Useful. PLOS Medicine.
- Harvard T.H. Chan School of Public Health. Types of Nutrition Studies and Their Limitations.
- U.S. Food & Drug Administration. Dietary Supplement Claims and Consumer Understanding.
- National Institutes of Health. How to Read and Evaluate Health Research.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Natural vs. Synthetic Vitamins: What the Evidence Actually Shows
An evidence-informed look at what “natural” and “synthetic” really mean in supplements—when the distinction matters, when it doesn’t, and how to think about these labels without getting misled.
A balanced look at natural and synthetic vitamins, and when the distinction meaningfully matters.
You are comparing two supplement labels. One says "natural" or "food-based." The other is described as "synthetic" somewhere in the marketing. The natural option may sound cleaner, safer, or more appropriate for a child. The synthetic option may sound like a compromise, even when the label does not explain why.
These reactions are understandable. "Natural" and "synthetic" are among the most used terms in the supplement space, and they carry strong associations. But they rarely explain how a vitamin behaves in the body, how it was formulated, or whether it meaningfully differs from an alternative source. ¹
For families trying to make thoughtful decisions, this framing can create unnecessary confusion. Products are marketed as superior based on origin alone, while more relevant factors, such as form, dose, and intended use, receive less attention.
This article examines what "natural" and "synthetic" mean in the context of vitamins, where the distinction can matter, and where it often does not. The goal is not to favor one category over the other, but to clarify how these terms are used, and often misused, so they can be evaluated appropriately.
Why This Question Comes Up So Often
Many supplement labels emphasize "natural" sourcing as a signal of quality, while other products are described as "synthetic" or "lab-made" in ways that imply inferiority. These distinctions are rarely explained beyond the label itself.
For parents, the concern is usually practical rather than philosophical. Is one safer than the other? Is one better absorbed? Is one more appropriate for children? Am I paying more for something that matters? Am I avoiding something unnecessarily?
The answers depend on context, the specific form used, and dose, not simply on whether an ingredient is described as natural or synthetic.¹
What the Natural-vs-Synthetic Label Suggests, and What It Does Not Prove
How This Fits Within an Evidence-First Approach
When evaluating supplements, the same core questions apply regardless of how an ingredient is described: What is the ingredient, chemically? What form is it in? How much is provided? What is the product meant to do? What tradeoffs are visible?
"Natural" and "synthetic" are descriptors of origin. They are not conclusions about quality, safety, or effectiveness.

This approach reflects the same evidence-first lens applied throughout this site, evaluating supplements consistently rather than relying on marketing shortcuts.
A one-page reference for reviewing ingredient identity, dose, form, formulation quality, and supplement claims more clearly.
View Checklist →What "Natural" Actually Means (and Doesn't)
In supplements, "natural" typically refers to an ingredient extracted from plants, animals, or microorganisms, or derived from a naturally occurring source before processing.
A natural-source label may describe where the ingredient started. It does not necessarily describe what the finished ingredient does in the body.
What the label does not automatically mean: gentler on the body, better absorbed, safer at higher doses, or free from processing or purification. Many natural-source ingredients require significant processing to isolate the active compound. In some cases, the final ingredient is chemically identical to a version produced through synthesis.² How supplement labels describe sourcing and processing often influences perception more than how the ingredient is used by the body.
What "Natural" Can Mean, and What It Does Not Guarantee
What "Synthetic" Actually Means
"Synthetic" generally refers to ingredients produced through controlled chemical processes and designed to replicate naturally occurring compounds.
Importantly, many synthetic vitamins are chemically identical to those found in food. Synthesis allows for consistency, stability, and precise dosing. Safety depends on quality controls and dose, not origin alone.
For some nutrients, synthetic forms are more extensively studied, easier to standardize, or more stable in supplement formulas.³ That does not make them universally superior, but it does mean that "synthetic" is not, on its own, a reason to avoid a product.
Origin is only one piece of the formulation story.
Whether a vitamin is natural or synthetic matters less than whether the specific form, dose, quality controls, and intended use make sense for the person taking it.
When Source Can Matter
There are situations where sourcing or formulation differences are relevant, but the reasons tend to be more specific than label language suggests.
Nutrients With Multiple Chemical Forms
Some vitamins exist in multiple forms that behave differently in the body. In these cases, the meaningful distinction is not "natural versus synthetic," but which specific form is used and how it is metabolized. That is a formulation question, not a sourcing one.
Whole-Food or Food-Based Blends
Some products use concentrated food extracts rather than isolated nutrients. These may include additional compounds from the source material, which can be relevant in some contexts. They can also introduce variability in composition and dosing, making it harder to evaluate what is being provided.
Sensitivity or Allergen Considerations
Certain natural sources, such as fish, yeast, or specific plant extracts, may be relevant for families managing sensitivities or avoiding particular ingredients. These are situational considerations, not a universal reason to prefer one category over the other.
In each case, source can matter. But it matters for specific, identifiable reasons, not because "natural" is broadly superior.
When source is part of the decision, ask:
Bioavailability Is About Form, Not Philosophy
Bioavailability is often cited as a reason to favor natural vitamins. In practice, absorption and utilization depend on the specific chemical form, the dose provided, interactions with other nutrients, and individual digestive and metabolic differences.⁴
Whether a vitamin is labeled natural or synthetic does not, on its own, determine how well it is absorbed or used by the body.⁴ Some synthetic forms are well absorbed and extensively studied. Some natural-source formulas offer no clear advantage beyond perception.
Discussions of bioavailability need to focus on specific forms and contexts rather than broad sourcing categories.
Bioavailability is a form-and-context question, not a natural-versus-synthetic shortcut.
A sourcing label does not tell you how well a nutrient will be absorbed or used. The specific chemical form, dose, formulation, and individual context matter more.
Where This Distinction Most Often Appears
Rather than evaluating individual brands, it is often more useful to recognize where the natural-versus-synthetic question commonly arises and why.
B vitamins, vitamin D, and vitamin C each exist in multiple forms with different metabolic pathways. In these cases, the form matters more than whether the source was natural or synthetic. Minerals are inorganic by nature, so differences typically relate to the compound they are bound to, not sourcing. Whole-food and food-based supplements emphasize where the ingredient came from, but they may trade dosing precision for sourcing appeal. And children's supplements frequently highlight natural flavors, colors, or sourcing, sometimes in ways that obscure meaningful formula differences.
Across all of these categories, source alone is an incomplete decision tool. This matters most in children's supplements, where simplified label language can create the impression of quality without revealing the details that support it.
Explore structured EFW resources for evaluating supplement tradeoffs, including sourcing, form, dosing precision, tolerance, and formulation clarity.
Explore the Frameworks Hub →Managing Expectations Around "Natural" Claims
"Natural" claims can create the impression that a product is inherently safer or more suitable for long-term use.⁵ That impression is worth examining. Safety is determined by dose, duration, quality controls, and individual factors, not by how an ingredient is labeled.
Assuming a product is risk-free because it is described as natural can lead to unnecessary supplementation or less careful attention to dosing, particularly in children. An evidence-first approach applies the same level of scrutiny to all supplements regardless of origin.
A "natural" claim should not lower the standard of evaluation.
Families can evaluate natural and synthetic products using the same core questions: What is the ingredient, what form is used, how much is provided, and what tradeoffs are visible?
Why This Matters for Families
For families, the natural-versus-synthetic question can become a distraction. It can lead parents to overpay for sourcing language without added benefit, avoid appropriate products unnecessarily, or overlook more important details like form, dose, quality testing, and tolerability.
When these labels are treated as absolutes, the decision shifts toward perception rather than evaluation. Understanding what "natural" and "synthetic" actually describe, and what they do not, helps redirect attention to the factors that matter most. The same evaluation questions used throughout this site apply equally to both categories.
Pulling It All Together
Natural and synthetic vitamins are descriptors of origin, not guarantees of effectiveness or safety. The most meaningful differences between supplements are rarely captured by those labels alone.
For families evaluating supplements, the most useful questions remain: Is supplementation appropriate right now? Does the specific form make sense for the intended use? Is the dose conservative and appropriate? Are expectations realistic and evidence-informed?
Answering these questions keeps decisions grounded in clarity rather than marketing. If you are building confidence evaluating supplements more broadly, the Evidence-Informed Supplement Evaluation Checklist can help organize the same questions across product categories.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- National Institutes of Health Office of Dietary Supplements. Dietary Supplements: What You Need to Know About Ingredients and Labels.
- Harvard T.H. Chan School of Public Health. Vitamins and Supplements: Understanding Forms and Bioavailability.
- National Institutes of Health Office of Dietary Supplements. Vitamin and Mineral Fact Sheets (Chemical Forms and Stability).
- Institute of Medicine (National Academies). Dietary Reference Intakes: Applications in Dietary Assessment. (Form and dose principles; accessible summaries via NIH/Harvard.)
- U.S. Food & Drug Administration. Dietary Supplement Labeling Guide.
All sources are freely accessible or summarized via NIH, FDA, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are referenced for educational comparison only, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
How to Evaluate Probiotics for Families: What Actually Matters
An evidence-informed framework for evaluating probiotics for families, focusing on strain relevance, formulation quality, and real-world use rather than broad claims or brand marketing.
A practical framework for assessing probiotic strains, dosing, and evidence across common use cases.
Probiotics are one of the most commonly recommended supplements for families, and one of the most confusing. A pediatrician suggests one after a course of antibiotics. A product label promises "gut health," "immune support," or "digestive balance." The strain names are long and unfamiliar. The CFU count looks impressive, but it is not obvious what that number actually means or whether higher is better.
For parents trying to make a thoughtful decision, this can quickly feel overwhelming, especially when the goal is simply to do something helpful without defaulting to a supplement indefinitely.
This article explains how to evaluate probiotics for families using an evidence-informed approach. Rather than focusing on brand claims or strain lists alone, it outlines what actually matters: purpose, strain relevance, formulation quality, and real-world practicality.
The goal is clarity, not complexity.
How Pediatric Guidance Views Probiotics
Unlike essential vitamins and minerals, probiotics are not considered universally necessary for healthy children.¹ Pediatric guidance generally frames them as context-dependent tools rather than routine daily supplements.
In practice, this means probiotics are most often considered during or after antibiotic use, in situations involving digestive disruption, or when families are looking for targeted gut support. They are not meant to replace a balanced diet or to be taken without a clear, defined reason.¹
Understanding this context helps set appropriate expectations from the start, and it shapes the most important question a parent can ask: not which probiotic looks strongest, but whether a probiotic is the right response to the situation at hand.
A probiotic should have a reason, not just a routine.
For families, the first question is not which probiotic looks strongest. It is whether a probiotic is appropriate for the situation, what role it is meant to play, and how long it should reasonably be used.
Step One: Clarify the Purpose
Before comparing products, the most useful question is: What job is this probiotic meant to do right now?
Is this related to digestive disruption after antibiotics? Is the goal short-term support during a specific period? Is it becoming a daily habit without a clearly defined reason? Is the intended use specific enough to evaluate whether the strains and format actually make sense?
Probiotics are strain-specific, meaning different strains are associated with different functions.² A product formulated for general digestive support may look very different from one intended to support gut recovery after antibiotics. Without a clear reason for use, it is difficult to evaluate whether a product's formula makes sense or whether its claims are meaningful.
This situational approach differs from how multivitamins are typically evaluated, which is discussed in Comparing Children's Multivitamins: An Evidence-Informed Guide for Parents.
Before comparing probiotic products, ask:
A short decision-support tool to help clarify whether supplement support may be worth considering and what type of question to ask next.
Start the Decision Tool →What Probiotics Are (and Why Strains Matter)
Probiotics are live microorganisms, most commonly bacteria, intended to support gut function. The most frequently used genera in children's products include Lactobacillus and Bifidobacterium.
Within each genus are individual strains, identified by a combination of letters and numbers. These identifiers are not marketing details. They distinguish organisms that can behave differently in the body, and the research behind one strain does not automatically apply to another.²
Two products can both be called children's probiotics and still be built around very different organisms, uses, and evidence. A label that lists only a genus name or a "probiotic blend" without strain-level identification does not give enough information to meaningfully evaluate the product.
What a Probiotic Label Can Tell You, and What Still Needs Context
Understanding CFUs Without Fixating on the Number
CFU stands for colony-forming units and reflects the number of live organisms at the time of manufacture or expiration, depending on how the product is formulated.
A high CFU count can look impressive on a label, but higher is not automatically better, particularly for children.³ The number only becomes meaningful when interpreted alongside the strains included, the intended purpose, the child's age and tolerance, how the product maintains stability through use, and the delivery format.
In pediatric formulas, CFU counts are often moderate and conservative by design, which aligns with safety-first principles.³ A lower count in a well-matched, stable formula may be more appropriate than a high count in a product with unclear strain relevance or poor viability.
A higher CFU count is not automatically a better probiotic.
CFU only becomes meaningful when interpreted alongside strain identity, intended purpose, delivery format, stability, and tolerance.
Practical Signals of Probiotic Quality
When evaluating probiotic products for families, looking beyond marketing language and toward a few practical quality signals makes comparisons more useful.
Clear strain identification matters most. Products that list organisms down to the strain level, not just the genus or species, offer greater transparency and allow for more meaningful evaluation of whether the product fits the intended use case.
Stability and storage information are also important signals. Because probiotics are living organisms, reputable products account for shelf life, storage conditions, and moisture exposure rather than relying on high CFU counts alone to suggest potency.⁴
A purpose-aligned formula is another indicator. A well-designed probiotic tends to have a clear use case rather than attempting to address every possible concern at once. Products that try to do everything for everyone are often harder to evaluate on any specific dimension.
For children especially, simplicity tends to be a strength. Formulas that avoid unnecessary additives, excess sweeteners, and large strain counts may be easier to tolerate and more realistic to use consistently.
Probiotic Evaluation Criteria: What to Check Before Choosing One
| Factor | Why It Matters | What to Look For | What to Be Careful About |
|---|---|---|---|
| Purpose | Probiotic evidence is often tied to specific outcomes or use cases | A clear reason for use, such as short-term digestive support or antibiotic-associated disruption | Broad "gut health" claims without a defined purpose |
| Strain identification | Effects can be strain-specific, not just genus- or species-wide | Full strain names or identifiers, not just "Lactobacillus" or "Bifidobacterium" | Products that list only general organism names |
| CFU count | The amount of live organisms matters, but only in context | CFU listed clearly, ideally through expiration when applicable | Assuming higher CFU automatically means better |
| Stability and storage | Probiotics are living organisms that may be affected by heat, moisture, and time | Clear storage instructions and shelf-life information | Vague viability claims or unclear storage requirements |
| Delivery format | Format affects dosing consistency, acceptance, and stability | A format that fits the child's age, routine, and tolerance | Choosing convenience while ignoring sweeteners, dose, or viability |
| Simplicity and tolerance | More strains or additives are not always better for children | Purposeful formula with unnecessary extras kept limited | Crowded formulas with unclear rationale |
This table is not a ranking system. It is a way to evaluate whether a probiotic's formula fits the intended purpose, child, and household context.
Why Formulation and Delivery Matter
A probiotic is not just an ingredient list. It is a living product that depends on stability, storage, delivery, and viability. This is why formulation and delivery play an especially important role in probiotics specifically, an idea explored more broadly in Why Dosage and Form Matter More Than Ingredient Lists.
Heat, moisture, and time can all affect whether the organisms in a probiotic remain viable by the time the product is used. Whether strains are clearly identified, how the product maintains stability through its shelf life, and whether it can realistically be stored in the household routine are all part of the evaluation.
Children's probiotics are commonly available as powders, chewables, liquids or drops, and gummies. Each format has tradeoffs related to taste, dosing consistency, and strain stability. No single format is right for every child or household.
A probiotic is not just an ingredient list. It is a living product.
Stability, storage, moisture exposure, delivery format, and strain viability can affect how a probiotic performs in real-world use.
Step Two: Understanding the Main Probiotic Categories for Families
Rather than comparing individual brands, it's often more useful to understand the major formulation categories used in children's probiotics. Each reflects different priorities around dosing, stability, convenience, and tolerance. Understanding these categories makes it easier to evaluate products on their design, not just their marketing.
A one-page reference for reviewing strain relevance, formulation quality, dosing, and supplement claims without relying on label language alone.
View Checklist →Common Probiotic Categories for Families
| Category | Often Prioritized For | Common Tradeoffs | Example Formulations |
|---|---|---|---|
| Single-strain probiotics | Specific situations; simplicity; children with sensitivities | Narrower scope; less general coverage | Culturelle Kids; Lactobacillus rhamnosus GG; Lactobacillus reuteri |
| Multi-strain products | General digestive support; broader formulation; short-term or intermittent use | Harder to assess strain relevance; more complex formulas; added sweeteners in gummy formats | Garden of Life Kids Probiotic Gummies |
| Shelf-stable probiotics | Travel, school use, convenience, consistent routines without refrigeration | Fewer strain options; formulation constraints to support stability | Align Kids |
| Refrigerated or stability-sensitive probiotics | Strain viability; controlled storage; powder-based delivery formats | Storage requirements; reduced convenience | SFI Health / Klaire Labs Children's Ther-Biotic |
| Minimalist / sensitive-friendly probiotics | Children with sensitivities; families reducing unnecessary additives; tolerance concerns | May still include multiple strains; narrower positioning; may not address multiple goals at once | Smidge Infant; Smidge Kids / Smidge Sensitive formulations |
| Powder or drop-based probiotics | Infants and young children; dosing flexibility; mixing with food or liquids | Taste masking; consistency of administration | BioGaia Baby Protectis Drops |
Example products are included for educational comparison only. They illustrate common formulation categories and are not rankings or recommendations.

Step Three: Evaluate Safety and Tolerance
Children's probiotics are typically formulated conservatively, but tolerance can still vary. Beyond the probiotic strains themselves, parents often consider sweeteners or flavoring agents in gummies and chewables, the delivery format and whether it fits the child's routine, and any prior history of digestive sensitivity that might affect how a new formula is tolerated.
For families, it is usually more useful to approach a new probiotic cautiously and pay attention to tolerance over the first few days, especially for younger children or those with a history of digestive sensitivity.⁵
Before using a probiotic routinely, ask:
Managing Expectations
Probiotic labels often use broad claims related to digestion, immunity, or overall wellness. Because the language can sound confident and comprehensive, it is easy to expect quick or dramatic results. That expectation is worth examining.
Probiotics can support gut function in specific contexts. They are not cure-alls, and they do not produce the same results across every child, situation, or product.⁵ When the expected effect does not appear quickly, some families switch products repeatedly, which rarely helps and can make evaluation harder.
Keeping expectations realistic, staying with a well-matched product long enough to observe a response, and building in a plan to reassess rather than continue indefinitely are all part of using probiotics thoughtfully.
A probiotic should be evaluated by fit, not expectation.
For families, the goal is not to find the strongest or most complex probiotic. The goal is to choose a product that fits the intended use, has a transparent formula, and can be used realistically for a defined reason.
How This Fits Within an Evidence-First Approach
Probiotics illustrate why a one-size-fits-all approach does not work. The same product may be appropriate in one situation, unnecessary in another, or poorly matched to a child's routine and tolerance.
This approach reflects the same evidence-first lens used throughout this site, explained in more detail in How I Evaluate Supplements as a Biomedical Engineer and Quality Professional.
Explore structured EFW resources for evaluating supplement tradeoffs, including strain relevance, dose, form, delivery format, quality signals, and real-world use.
Explore the Frameworks Hub →Why This Matters for Families
Probiotic decisions often come up when a child is uncomfortable or recovering from disruption: illness, antibiotic use, digestive discomfort. In those moments, parents naturally want to do something helpful. That instinct is reasonable. But the more useful question is not which probiotic looks strongest. It is what the probiotic is meant to help with, and whether the product in hand actually fits the situation.
Having a clearer way to evaluate products, starting with the reason for use and working through strain relevance, CFU in context, stability, format, and tolerance, helps reduce guesswork and the pressure to find a "best" option that may not exist for every situation.
Pulling It All Together
A probiotic decision is strongest when it starts with a clear reason for use, then checks whether the strains are relevant to that situation, whether the CFU count is meaningful in context, whether the product is stable and stored appropriately, whether the format fits the child and household routine, and whether the expectations are realistic and time-limited.
That sequence of questions matters more than the brand name or the size of the CFU number on the label. For families, it is also what makes the decision feel grounded rather than guesswork.
This way of evaluating probiotics aligns with the broader supplement evaluation approach outlined in How I Evaluate Supplements as a Biomedical Engineer and Quality Professional. For families still clarifying whether a probiotic is the right step, the Evidence-Informed Supplement Evaluation Checklist walks through the same evaluation steps in a practical format.
Browse side-by-side comparison content designed to help families evaluate supplement categories through formulation, dose, quality signals, and tradeoffs.
View All Comparisons →Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- American Academy of Pediatrics. Probiotics and Prebiotics in Pediatrics.
- National Institutes of Health Office of Dietary Supplements. Probiotics Fact Sheet for Health Professionals.
- Hill C et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of probiotics. Nature Reviews Gastroenterology & Hepatology.
- National Institutes of Health Office of Dietary Supplements. Probiotics: Health Professional Fact Sheet (Stability and Viability sections).
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are included as educational examples based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
How I Evaluate Supplements as a Biomedical Engineer and Quality Professional
A biomedical engineer explains how to evaluate supplements using an evidence-first, quality-driven framework—focused on ingredients, formulation, risk, and real-world use for families.
A structured look at how supplements are assessed through an evidence-, safety-, and quality-centered lens.
Choosing a supplement shouldn't feel like you're navigating a marketing maze. Yet, for many families, finding a clear, straightforward answer is a constant struggle.
You're standing in a store aisle comparing two bottles with nearly identical claims, or scrolling through product pages where all brands sound scientifically credible. Someone you trust recommended one thing, while another source recommends something contradictory. You're not trying to become a supplement expert. You're just trying to make a reasonable decision for your child.
The supplement space doesn't make it easy. Labels sound scientific, claims feel convincing, and the advice frequently contradicts itself in ways that are genuinely hard to sort through without a framework for what actually matters.
I evaluate supplements professionally, but my approach isn't about tracking brands or following trends. It's about understanding how something works in the body, whether it is likely to do what it claims, and whether it makes sense for the specific person considering it. Those are different questions, and they lead to different answers.
This article explains how I think about supplements, and how you can start asking the same fundamental questions, even without a science background.
What "Evidence-Based" Actually Means (and What It Doesn't)
"Evidence-based" is one of the most overused phrases in wellness. Used loosely enough, it starts to mean almost nothing.
When I use the term, I'm not describing products with testimonials, trending ingredients, or claims that sound scientific. A study mentioned in a marketing email is not evidence-based thinking, and neither is a familiar ingredient name. Those signals can feel reassuring. That is not enough.
Evidence-based thinking starts down a different path: Is there biological plausibility? Is the dose meaningful? Is the form appropriate for how the body actually absorbs and uses this ingredient? Is the research relevant to the person taking it, or was it conducted in a different population, at a different dose, under different conditions? Context can completely change the meaning of the evidence.
Evidence-based assessment also means being comfortable with genuine uncertainty. Not every supplement has robust data, and that alone does not make it ineffective, but it does change how cautiously it should be approached. Acknowledging that gap is part of what evidence-based reasoning actually looks like.
How evidence is selected, interpreted, and cited across this site is explained in How We Cite Evidence.
What Sounds Evidence-Based, and What Actually Needs to Be Evaluated
Why Supplement Claims Require a Different Kind of Scrutiny
Many people assume that if a supplement is sold legally, its claims have been evaluated in the same way as those of pharmaceutical products. That's not quite how the system works, and understanding the difference matters more than most labels let on.
In the United States, dietary supplements are regulated by the FDA, including oversight of manufacturing practices, ingredient safety, and labeling requirements. But unlike prescription or over-the-counter drugs (OTCs), supplement claims are not reviewed or approved by the FDA before products reach the market, as long as those claims do not cross into the realm of diagnosing, treating, preventing, or curing disease.
Instead, companies are permitted to make structure/function claims, which are statements about supporting normal bodily processes or maintaining general health. The strength of evidence behind those claims can vary enormously. As long as they stay within that framework, they do not go through the same pre-market evaluation required for pharmaceuticals.
This is why familiar supplement language can sometimes feel more specific than it actually is. For instance, a product may say it "supports immune health" or "helps maintain normal digestive regularity." Those phrases may sound reassuring, but they are still broad structure/function claims that do not necessarily tell you whether the product has been shown to meaningfully improve an outcome for a specific person, at a specific dose, in a specific context.
This does not mean that supplements are unregulated, but it does mean that the responsibility for interpreting claims falls largely on the consumer. Without a way to assess biological plausibility, dosage, formulation, and context, it is easy to read scientific-sounding language and assume it reflects pharmaceutical-level evidence when it does not. A claim can be legally compliant and still leave a family without enough information to make an informed decision.
That's the gap this framework is designed to close.
A claim can be legal and compliant yet still incomplete for decision-making.
Supplement claims can describe intended support without proving that a product is well-matched to a specific person, dose, or context. That is why evidence, formulation, and risk have to be evaluated together.
The Lens I Use When Evaluating Supplements
I've spent my professional life thinking about systems: how inputs, controls, and processes interact to produce real-world outcomes, and where things go wrong when they don't. That background shapes how I view supplement claims.
My training is in biomedical engineering. My day-to-day work is in quality systems for regulated manufacturing environments, where I lead oversight across multiple sites, ensuring products perform as intended under actual use conditions. In that world, what looks acceptable on paper is not always what happens in practice. Small details matter. Variability is real. Tradeoffs and failure points exist at every stage.
That same attention to inputs, controls, consistency, risk, and tradeoffs does not turn off when I evaluate a supplement. Instead of asking "Is this ingredient good?", I'm asking: How is this supposed to work biologically? What does the formulation assume? And where is the likely gap between the claim and the real-world outcome?
I'm not just interested in whether evidence exists. I'm trained to ask whether a product will actually do what it is supposed to do: reliably, in the hands of a real person, under real conditions. That's a different standard. And it's a more useful one for families trying to make decisions that are based on more than the claim on the label.
The goal is not skepticism for its own sake. It is better fit and better reasoning.
A supplement should be evaluated by how well its evidence, formulation, dose, and context align with the person and purpose it is meant to support.
Use the checklist to review ingredient purpose, dose, form, quality signals, and population context before relying on supplement claims.
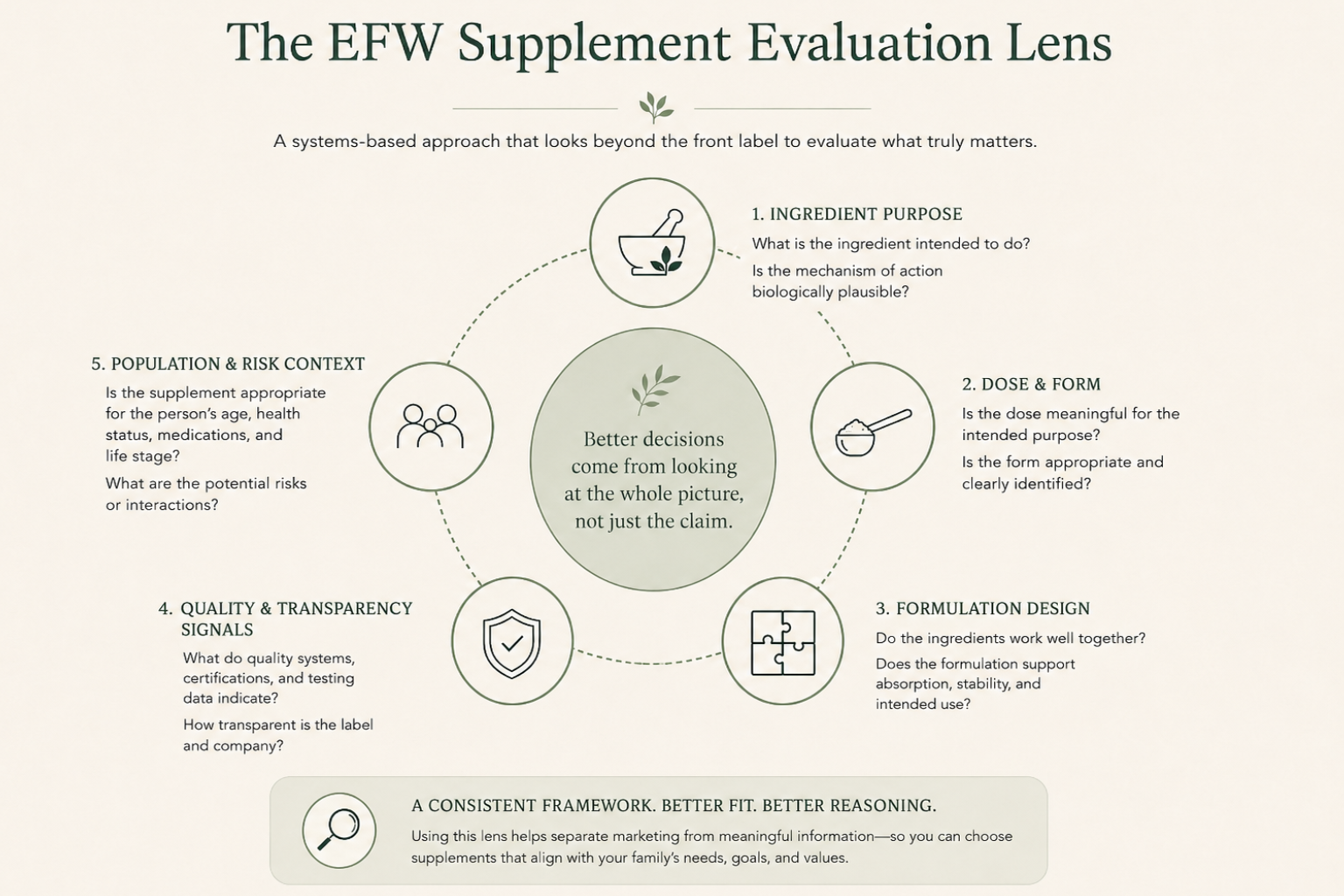
View the Checklist →The Five Things I Look at When Evaluating a Supplement
1. Ingredient Identity and Purpose
The first question is simple: Why is this ingredient here? Many supplements contain ingredients included for marketing appeal, present at doses that may be too low to be meaningful, or loosely matched to the stated purpose at best. I look for a clear connection between the ingredient, its known biological roles, and the claim being made. If that connection requires a lot of indirect reasoning to hold together, or disappears entirely when you look closely, it's a signal to slow down before assuming the product does what the label implies.
2. Dosage and Form Matter More Than Most People Realize
An ingredient's name tells you very little on its own. Dosage matters. Form matters. A supplement can include a well-studied ingredient and still be unlikely to produce meaningful effects if the dose is too low, the form is poorly absorbed, or it competes with other components in the formulation. This is one of the most common gaps between how supplements are marketed and how they actually perform. It rarely appears on the front label.
3. Formulation Quality Is Often Overlooked
Supplements are not just collections of ingredients dropped into a capsule. They are formulations. How ingredients interact, how stable they remain over time, and how they are delivered all affect whether a product performs as intended. A formula can look reasonable on paper. If it has not been designed thoughtfully as a system, performance suffers in ways that ingredient lists alone will not reveal.
4. Label Transparency Tells You a Lot
A label cannot tell you everything, but it tells you more than many people notice. I look at whether ingredients are clearly identified, whether doses are disclosed, whether claims are specific or vague, and what is conspicuously missing. Broad claims, proprietary blends, and ambiguous language are consistent patterns in products where marketing considerations have outweighed clarity. Transparency does not guarantee quality. But a lack of it should raise questions.
5. Risk and Population Context Are Non-Negotiable
A supplement that may be reasonable for a healthy adult is not automatically appropriate for children, pregnant or breastfeeding women, people taking medications, or people with underlying health conditions. I always consider who a supplement is intended for, who it may not be appropriate for, and what level of caution is appropriate. For families, this is not a footnote. It is a primary filter.
Before trusting a supplement claim, ask:
What I Don't Prioritize (On Purpose)
Brand popularity, influencer recommendations, trend status, and undefined "clean" or "natural" labels are often the first things people encounter when shopping for supplements. That's understandable. They're visible, they're designed to be persuasive, and there is usually a ready explanation attached to all of them.
I'm not dismissing parents who have started there. Most people do. The problem is not that these signals exist. They are not a substitute for understanding whether a product actually works, and whether it makes sense for the specific person using it. Starting with them, rather than returning to them later, tends to produce decisions driven more by marketing than by evidence.
What I Do Not Start With, and What I Look At First
Why This Matters for Families
When you're making a supplement decision for yourself, uncertainty feels different than when you're making it for a child. Not because every choice is high-stakes (most aren't), but because parents are rarely asking "Does this sound good?" They're asking: Is this necessary? Is it safe for my child specifically? Is it likely to help? Is it worth it?
Those are harder questions, and the supplement category does not always make them easier to answer. Parents are frequently navigating incomplete information, competing advice from sources they trust, and a true desire not to overreact or underthink. That is not a failure of judgment. It is a reasonable response to a category that is genuinely difficult to evaluate without a clear framework.
The goal here is not perfect certainty. It is a better set of starting questions, ones that get past the marketing layer and into what actually affects whether a supplement is the right fit.
Better supplement decisions do not require perfect certainty. They require a better set of questions.
For families, the goal is not to eliminate every uncertainty. It is to avoid relying on marketing shortcuts when evidence, dose, formulation, and context provide a clearer way to think.
How to Use This Framework Going Forward
Asking better questions does not require a science background. It requires knowing what to look for and then practicing the habit of looking for it. As you read content on this site, you'll see this same framework applied across specific supplements and decisions: What is the ingredient? What is the dose? How is it formulated? Who is it for? What are the tradeoffs?
The Frameworks & Decision Tools Hub applies this lens across supplement categories in more depth. Over time, this way of thinking becomes more intuitive, and the category starts to feel less overwhelming, because you know what you're actually trying to figure out.
A deeper, structured guide to evaluating multivitamins, probiotics, and magnesium using the same evidence-first framework introduced in this article.
Learn More →New articles and practical frameworks are also delivered weekly through the Evidence First Digest.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Some content may include affiliate links to products that meet Evidence First Wellness evaluation criteria. If you choose to purchase through those links, Evidence First Wellness may earn a commission at no additional cost to you. Affiliate relationships do not determine editorial conclusions.
References and Further Reading
Foundational concepts discussed here are informed by:
- Nutritional biochemistry and pharmacokinetics literature
- Regulatory guidance related to dietary supplements
- Peer-reviewed research on formulation science and bioavailability
Specific sources are referenced in ingredient- and product-focused articles where they are most directly relevant.







