How I Evaluate Supplements as a Biomedical Engineer and Quality Professional
A structured look at how supplements are assessed through an evidence-, safety-, and quality-centered lens.
Choosing a supplement shouldn't feel like you're navigating a marketing maze. Yet, for many families, finding a clear, straightforward answer is a constant struggle.
You're standing in a store aisle comparing two bottles with nearly identical claims, or scrolling through product pages where all brands sound scientifically credible. Someone you trust recommended one thing, while another source recommends something contradictory. You're not trying to become a supplement expert. You're just trying to make a reasonable decision for your child.
The supplement space doesn't make it easy. Labels sound scientific, claims feel convincing, and the advice frequently contradicts itself in ways that are genuinely hard to sort through without a framework for what actually matters.
I evaluate supplements professionally, but my approach isn't about tracking brands or following trends. It's about understanding how something works in the body, whether it is likely to do what it claims, and whether it makes sense for the specific person considering it. Those are different questions, and they lead to different answers.
This article explains how I think about supplements, and how you can start asking the same fundamental questions, even without a science background.
What "Evidence-Based" Actually Means (and What It Doesn't)
"Evidence-based" is one of the most overused phrases in wellness. Used loosely enough, it starts to mean almost nothing.
When I use the term, I'm not describing products with testimonials, trending ingredients, or claims that sound scientific. A study mentioned in a marketing email is not evidence-based thinking, and neither is a familiar ingredient name. Those signals can feel reassuring. That is not enough.
Evidence-based thinking starts down a different path: Is there biological plausibility? Is the dose meaningful? Is the form appropriate for how the body actually absorbs and uses this ingredient? Is the research relevant to the person taking it, or was it conducted in a different population, at a different dose, under different conditions? Context can completely change the meaning of the evidence.
Evidence-based assessment also means being comfortable with genuine uncertainty. Not every supplement has robust data, and that alone does not make it ineffective, but it does change how cautiously it should be approached. Acknowledging that gap is part of what evidence-based reasoning actually looks like.
How evidence is selected, interpreted, and cited across this site is explained in How We Cite Evidence.
What Sounds Evidence-Based, and What Actually Needs to Be Evaluated
Why Supplement Claims Require a Different Kind of Scrutiny
Many people assume that if a supplement is sold legally, its claims have been evaluated in the same way as those of pharmaceutical products. That's not quite how the system works, and understanding the difference matters more than most labels let on.
In the United States, dietary supplements are regulated by the FDA, including oversight of manufacturing practices, ingredient safety, and labeling requirements. But unlike prescription or over-the-counter drugs (OTCs), supplement claims are not reviewed or approved by the FDA before products reach the market, as long as those claims do not cross into the realm of diagnosing, treating, preventing, or curing disease.
Instead, companies are permitted to make structure/function claims, which are statements about supporting normal bodily processes or maintaining general health. The strength of evidence behind those claims can vary enormously. As long as they stay within that framework, they do not go through the same pre-market evaluation required for pharmaceuticals.
This is why familiar supplement language can sometimes feel more specific than it actually is. For instance, a product may say it "supports immune health" or "helps maintain normal digestive regularity." Those phrases may sound reassuring, but they are still broad structure/function claims that do not necessarily tell you whether the product has been shown to meaningfully improve an outcome for a specific person, at a specific dose, in a specific context.
This does not mean that supplements are unregulated, but it does mean that the responsibility for interpreting claims falls largely on the consumer. Without a way to assess biological plausibility, dosage, formulation, and context, it is easy to read scientific-sounding language and assume it reflects pharmaceutical-level evidence when it does not. A claim can be legally compliant and still leave a family without enough information to make an informed decision.
That's the gap this framework is designed to close.
A claim can be legal and compliant yet still incomplete for decision-making.
Supplement claims can describe intended support without proving that a product is well-matched to a specific person, dose, or context. That is why evidence, formulation, and risk have to be evaluated together.
The Lens I Use When Evaluating Supplements
I've spent my professional life thinking about systems: how inputs, controls, and processes interact to produce real-world outcomes, and where things go wrong when they don't. That background shapes how I view supplement claims.
My training is in biomedical engineering. My day-to-day work is in quality systems for regulated manufacturing environments, where I lead oversight across multiple sites, ensuring products perform as intended under actual use conditions. In that world, what looks acceptable on paper is not always what happens in practice. Small details matter. Variability is real. Tradeoffs and failure points exist at every stage.
That same attention to inputs, controls, consistency, risk, and tradeoffs does not turn off when I evaluate a supplement. Instead of asking "Is this ingredient good?", I'm asking: How is this supposed to work biologically? What does the formulation assume? And where is the likely gap between the claim and the real-world outcome?
I'm not just interested in whether evidence exists. I'm trained to ask whether a product will actually do what it is supposed to do: reliably, in the hands of a real person, under real conditions. That's a different standard. And it's a more useful one for families trying to make decisions that are based on more than the claim on the label.
The goal is not skepticism for its own sake. It is better fit and better reasoning.
A supplement should be evaluated by how well its evidence, formulation, dose, and context align with the person and purpose it is meant to support.
Use the checklist to review ingredient purpose, dose, form, quality signals, and population context before relying on supplement claims.
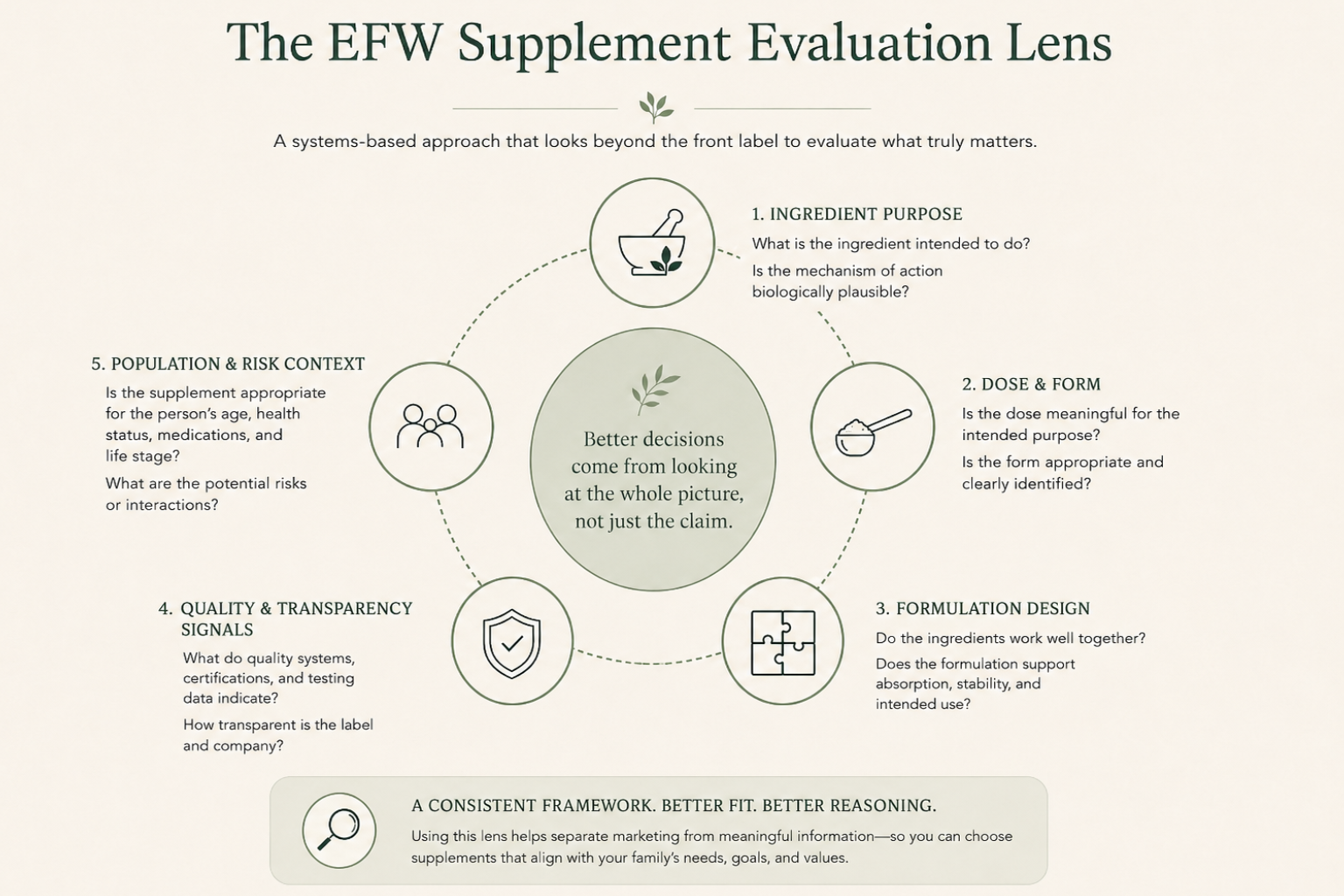
View the Checklist →The Five Things I Look at When Evaluating a Supplement
1. Ingredient Identity and Purpose
The first question is simple: Why is this ingredient here? Many supplements contain ingredients included for marketing appeal, present at doses that may be too low to be meaningful, or loosely matched to the stated purpose at best. I look for a clear connection between the ingredient, its known biological roles, and the claim being made. If that connection requires a lot of indirect reasoning to hold together, or disappears entirely when you look closely, it's a signal to slow down before assuming the product does what the label implies.
2. Dosage and Form Matter More Than Most People Realize
An ingredient's name tells you very little on its own. Dosage matters. Form matters. A supplement can include a well-studied ingredient and still be unlikely to produce meaningful effects if the dose is too low, the form is poorly absorbed, or it competes with other components in the formulation. This is one of the most common gaps between how supplements are marketed and how they actually perform. It rarely appears on the front label.
3. Formulation Quality Is Often Overlooked
Supplements are not just collections of ingredients dropped into a capsule. They are formulations. How ingredients interact, how stable they remain over time, and how they are delivered all affect whether a product performs as intended. A formula can look reasonable on paper. If it has not been designed thoughtfully as a system, performance suffers in ways that ingredient lists alone will not reveal.
4. Label Transparency Tells You a Lot
A label cannot tell you everything, but it tells you more than many people notice. I look at whether ingredients are clearly identified, whether doses are disclosed, whether claims are specific or vague, and what is conspicuously missing. Broad claims, proprietary blends, and ambiguous language are consistent patterns in products where marketing considerations have outweighed clarity. Transparency does not guarantee quality. But a lack of it should raise questions.
5. Risk and Population Context Are Non-Negotiable
A supplement that may be reasonable for a healthy adult is not automatically appropriate for children, pregnant or breastfeeding women, people taking medications, or people with underlying health conditions. I always consider who a supplement is intended for, who it may not be appropriate for, and what level of caution is appropriate. For families, this is not a footnote. It is a primary filter.
Before trusting a supplement claim, ask:
What I Don't Prioritize (On Purpose)
Brand popularity, influencer recommendations, trend status, and undefined "clean" or "natural" labels are often the first things people encounter when shopping for supplements. That's understandable. They're visible, they're designed to be persuasive, and there is usually a ready explanation attached to all of them.
I'm not dismissing parents who have started there. Most people do. The problem is not that these signals exist. They are not a substitute for understanding whether a product actually works, and whether it makes sense for the specific person using it. Starting with them, rather than returning to them later, tends to produce decisions driven more by marketing than by evidence.
What I Do Not Start With, and What I Look At First
Why This Matters for Families
When you're making a supplement decision for yourself, uncertainty feels different than when you're making it for a child. Not because every choice is high-stakes (most aren't), but because parents are rarely asking "Does this sound good?" They're asking: Is this necessary? Is it safe for my child specifically? Is it likely to help? Is it worth it?
Those are harder questions, and the supplement category does not always make them easier to answer. Parents are frequently navigating incomplete information, competing advice from sources they trust, and a true desire not to overreact or underthink. That is not a failure of judgment. It is a reasonable response to a category that is genuinely difficult to evaluate without a clear framework.
The goal here is not perfect certainty. It is a better set of starting questions, ones that get past the marketing layer and into what actually affects whether a supplement is the right fit.
Better supplement decisions do not require perfect certainty. They require a better set of questions.
For families, the goal is not to eliminate every uncertainty. It is to avoid relying on marketing shortcuts when evidence, dose, formulation, and context provide a clearer way to think.
How to Use This Framework Going Forward
Asking better questions does not require a science background. It requires knowing what to look for and then practicing the habit of looking for it. As you read content on this site, you'll see this same framework applied across specific supplements and decisions: What is the ingredient? What is the dose? How is it formulated? Who is it for? What are the tradeoffs?
The Frameworks & Decision Tools Hub applies this lens across supplement categories in more depth. Over time, this way of thinking becomes more intuitive, and the category starts to feel less overwhelming, because you know what you're actually trying to figure out.
A deeper, structured guide to evaluating multivitamins, probiotics, and magnesium using the same evidence-first framework introduced in this article.
Learn More →New articles and practical frameworks are also delivered weekly through the Evidence First Digest.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Some content may include affiliate links to products that meet Evidence First Wellness evaluation criteria. If you choose to purchase through those links, Evidence First Wellness may earn a commission at no additional cost to you. Affiliate relationships do not determine editorial conclusions.
References and Further Reading
Foundational concepts discussed here are informed by:
- Nutritional biochemistry and pharmacokinetics literature
- Regulatory guidance related to dietary supplements
- Peer-reviewed research on formulation science and bioavailability
Specific sources are referenced in ingredient- and product-focused articles where they are most directly relevant.

