When “More Is Better” Becomes Risky in Supplement Use
A clear explanation of how excessive dosing can introduce risk, and where upper limits begin to matter.
A label says "extra strength," "high potency," or "maximum support." You are comparing a standard dose to a stronger version and wondering whether the higher option offers more protection. A child is already taking a multivitamin, getting nutrients from fortified foods, and maybe a gummy on top of that. Choosing the stronger product can feel like covering all the bases.
That instinct is understandable. But with nutrients, more only helps up to a point. Once adequacy is reached, the question changes from whether more will help to whether more adds unnecessary exposure.
This article explores when increased intake may be justified, when it becomes unnecessary, and how to think about dose, safety margins, and cumulative exposure in a more balanced way.
The goal is proportionality, not fear.
Why "More Is Better" Feels Intuitive
The logic can feel reasonable: if too little is harmful, more must be safer. Supplements are associated with health support, deficiency is framed as consequential, and labels often emphasize potency as a mark of quality. Online advice can blur the line between meeting needs and doing more.
The problem is that nutrition does not always work that way. Higher doses can feel like greater protection without adding meaningful benefit once needs are already met. Understanding where that assumption breaks down is the more useful starting point.
How This Fits Within an Evidence-First Approach
Across this site, supplements are evaluated as tools designed to meet specific needs, not as performance enhancers. The useful question here is not "Which option is strongest?" It is "What dose fits the need without adding unnecessary exposure?"
This article uses the same evidence-first lens to examine dosing decisions, focusing on adequacy, safety thresholds, and current need rather than defaulting to higher intake. For a broader look at when supplementation may be worth considering at all, see How to Decide Whether a Supplement Is Worth Trying at All and Do Children Actually Need Supplements?
When Adequacy Is the Goal
Nutrient recommendations are generally designed to achieve adequacy, the level sufficient to prevent deficiency in most people. Moving beyond adequacy does not automatically produce additional benefit. In many cases, benefits plateau once sufficiency is reached, excess intake is excreted or accumulates depending on the nutrient, and risk begins to increase before additional benefit does.²
This is especially relevant in children, where margins between adequate and excessive intake may be narrower than in adults.
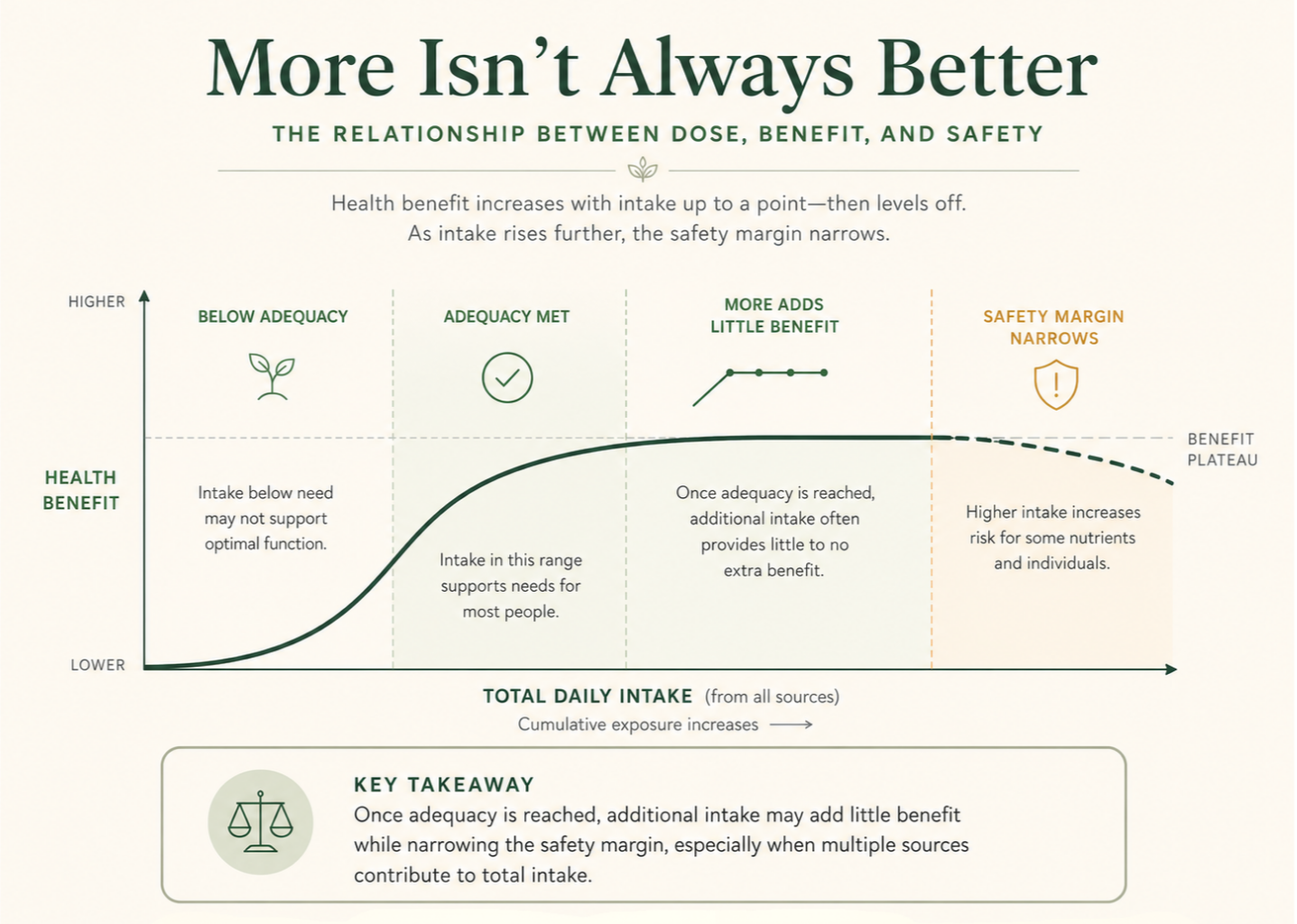
Upper Intake Levels and Safety Margins
For many nutrients, regulatory and scientific bodies establish tolerable upper intake levels (ULs).³ These levels are not ideal targets. They are thresholds beyond which the risk of adverse effects may increase.⁴ A number below the upper limit does not automatically mean the dose is useful, and a number near the upper limit should not be treated as a goal.
Long-term intake near or above these thresholds, particularly when multiple products are combined, can unintentionally reduce safety margins. Understanding that ULs are safety boundaries helps reframe what "high dose" means in practice.
Children Are Not Small Adults
Children differ from adults in body mass, metabolic rate, developmental stage, and nutrient requirements, all of which influence how nutrients are processed and tolerated.⁵ Higher doses that may be tolerated in adults do not automatically translate to pediatric safety.
This is explored in more depth in the broader discussion of pediatric versus adult supplement guidance, which covers why age-specific dosing and evidence matter independently of what works in adult populations.
When Higher Doses May Be Appropriate
There are situations where higher-than-baseline doses are used intentionally, including medically identified deficiencies, therapeutic short-term use, and specific clinical guidance.⁶ In these cases, increased intake is typically time-limited, monitored, and based on identified need. The key difference is intentionality, defined need, and duration, not potency alone.
Cumulative Exposure Is Often Overlooked
A child may not be taking a "high dose" from any single product. But the total can change quickly when a multivitamin, a gummy, fortified foods, and a single-nutrient supplement are all part of the routine. Each source contributes to total daily intake, and without periodic reassessment, cumulative intake can drift upward without anyone noticing.⁷
Form and dose tend to matter more than ingredient count alone, particularly when intake adds up across products. How to Read a Supplement Label (Without Getting Misled) explains how to identify overlapping nutrients across the products a family is already using.
Marketing and the Language of Potency
Labels often use "extra strength," "high potency," or "maximum support" as shorthand for a product being better or more protective. These phrases imply greater benefit but rarely clarify whether higher dosing is necessary or appropriate for the situation. Potency can sound like value, but value depends on whether the dose fits the need.
The Plateau Effect
A nutrient can be essential and still have a point where more stops helping. In nutritional science, many nutrients demonstrate a plateau effect: once physiological needs are met, additional intake does not improve outcomes.⁸ Continuing to increase intake beyond this plateau may add cost and complexity, reduce safety margins over time, and create a false sense of security.
For some nutrients, particularly those that accumulate in the body or come from multiple overlapping sources, higher intake can gradually narrow the gap between adequacy and excess. Recognizing this pattern helps counteract the assumption that a higher dose is a safer dose.
When More Stops Adding Value
| Dose Range | What It May Mean | Decision Lens |
|---|---|---|
| Below adequacy | Intake may not be sufficient to meet physiological needs | Evaluate dietary sources first; targeted supplementation may be appropriate |
| At adequacy | Needs are likely met; additional intake is unlikely to add meaningful benefit | Maintain without increasing; reassess if diet or situation changes |
| Above adequacy | Intake exceeds typical needs; benefit plateau has likely been reached | Question whether higher dose is necessary; monitor for cumulative exposure |
| Approaching upper limits | Safety margin is narrowing; especially relevant with combined supplement use | Review all sources of intake; dose reduction or product consolidation may be warranted |
| High-dose use | Risk of adverse effects may increase; particularly relevant in children and long-term use | Use only with clear clinical reason, defined duration, and professional oversight |
Increasing dose adds value only up to adequacy. Beyond that, safety margins may narrow.
A Practical Way to Evaluate Dose
Instead of asking "Is this the strongest option?" it can help to ask: What is the identified need? Is current intake already adequate? Does increasing the dose meaningfully reduce risk? Is higher dosing temporary or indefinite?
This reframing keeps the focus on whether the dose is useful, necessary, and proportionate. The Frameworks & Decision Tools Hub includes structured tools that apply this same thinking across different supplement categories.
The Evidence-Informed Family Supplement Guide is coming soon. It will walk through dosing, label review, cumulative exposure, safety margins, and category-specific tradeoffs for evaluating family supplement decisions with more clarity.
See What's Coming →Preventing Dose Drift
Dose drift happens when a supplement is increased or layered on out of caution, but the reason for the higher dose is never clearly defined. The starting point makes sense, the habit continues, and the dose becomes part of the routine without periodic review. The Supplement Decision Flow walks through adequacy, safety margins, cumulative exposure, and duration before adjusting dose upward.
Use the decision flow to work through adequacy, safety margins, cumulative exposure, and duration before adjusting dose upward. No rankings. No performance framing.
Use the Supplement Decision Flow →Why This Matters for Families
Parents often choose higher-dose products from a place of care. It can feel responsible to pick the stronger option, especially when labels suggest it provides more support. But if a child's needs are already met, a higher dose may not add more protection. It may simply add more exposure, especially when products are being combined.
Understanding where benefit plateaus and where safety margins narrow supports calmer, more proportionate decisions. The instinct to cover all bases is understandable. The goal is to make sure "covering all bases" means meeting the need, not exceeding it.
Pulling It All Together
Once adequacy is achieved, increasing intake does not guarantee additional benefit and may reduce safety margins over time. An evidence-informed approach prioritizes adequacy over potency, defined need over habit, and periodic review over automatic continuation.
A supplement dose is most useful when it addresses a defined need without adding unnecessary exposure. Once needs are met, the strongest option is not automatically the best-matched one. The most useful question is not "Is this the strongest option?" but whether this dose meaningfully addresses the need, or simply increases exposure.
Use the checklist to review ingredient identity, dosing, formulation quality, and supplement claims before deciding whether a product and dose fit the need.
View the Checklist →Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- Heaney RP. Nutrient Dose-Response Relationships and the Plateau Effect. American Journal of Clinical Nutrition.
- Institute of Medicine (National Academies). Dietary Reference Intakes: Applications in Dietary Assessment.
- National Academies of Sciences, Engineering, and Medicine. Tolerable Upper Intake Levels for Vitamins and Minerals.
- National Academies of Sciences, Engineering, and Medicine. Risk Assessment Model for Establishing ULs.
- National Institutes of Health. Pediatric Research and Age-Specific Dosing Principles.
- National Institutes of Health, Office of Dietary Supplements. Iron, Vitamin D, and Fat-Soluble Vitamin Fact Sheets.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know About Safety.
- Hathcock JN. Risk Assessment for Vitamins and Minerals. American Journal of Clinical Nutrition.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.

