Articles
These articles are designed to help families make thoughtful, evidence-informed decisions about supplement, skincare, and everyday wellness.
Rather than focusing on trends or product recommendations, each piece explains how to evaluate ingredients, formulation quality, label claims, and risk—so decisions feel clearer and more grounded.
Each article stands on its own wile contributing to a broader framework focused on clarity, safety, and real-world practicality.
How Pediatric Supplement Guidance Differs From Adult Recommendations
Children are not simply smaller adults. From dosing thresholds to safety considerations and clinical evidence, pediatric supplement guidance follows distinct principles. This article outlines where those differences matter — and why they exist.
A structured comparison of how supplement guidance shifts between children and adults, and why those differences exist.
A podcast discusses an adult supplement routine. A parent sees a higher-dose product and wonders whether a smaller amount of the same thing would work for a child. A children's multivitamin looks much lower in dose than an adult version and the difference is not explained. Adult supplement habits are framed around energy, performance, stress, or immunity, and it is not always clear what applies to children.
The lack of clear distinctions between adult and pediatric guidance can make this genuinely difficult to sort through. Supplement guidance often appears universal, and dosage language can feel interchangeable across age groups. But pediatric recommendations differ meaningfully from adult guidance, both in principle and in practice.
Children are not simply smaller adults.¹ Their physiology, developmental stage, safety margins, and evidence base all shape how supplement decisions should be made. A children's supplement that looks less impressive than its adult counterpart is not necessarily weaker. It may simply reflect a different set of priorities.
This article explores how pediatric supplement guidance differs from adult recommendations, why those differences exist, and how families can approach decisions through a pediatric lens.
The goal is proportionality, not restriction.
Why This Distinction Matters
The issue is not that adult supplement habits are always wrong. The issue is that they are built around a different body, different goals, and a different risk tolerance. Many supplement practices originate in adult wellness culture: higher doses, performance framing, enhancement goals, and routine long-term use. When those habits are applied to children without adjustment, they can create dosing misalignment, overestimation of need, inappropriate extrapolation of adult research, and unnecessary exposure.
Understanding how pediatric guidance differs helps prevent well-intentioned but mismatched decisions. For a broader look at when supplementation may or may not be appropriate for children, see Do Children Actually Need Supplements?
How This Fits Within an Evidence-First Approach
Across this site, supplements are treated as conditional tools, not defaults. For children, that lens becomes even more important because dose, duration, evidence, and safety margins all change with age and development.
This article uses that same evidence-first approach to compare pediatric and adult supplementation decisions, with attention to adequacy, safety margins, and age-specific need.
Children Are Not Smaller Adults
| Decision Area | Children's Supplement Decisions | Adult Supplement Decisions | Why It Matters |
|---|---|---|---|
| Dose | Age-stratified; conservative; calibrated to body mass and development | Often based on population averages; may emphasize higher potency | Pediatric doses do not scale linearly from adult doses |
| Safety margin | Narrower; precaution is emphasized; upper intake levels are age-specific | Wider in most cases; adults may accept voluntary tradeoffs at higher doses | Excess intake in children may reach concerning thresholds more quickly |
| Evidence base | Limited; often extrapolated cautiously from adult data | More extensive; longer-term and larger studies more common | Adult findings do not automatically translate to pediatric safety or efficacy |
| Duration | Typically short-term or purpose-driven; periodic reassessment encouraged | Long-term routines more common; continuation often assumed | Cumulative exposure in children warrants more active monitoring |
| Risk tolerance | Lower; threshold for justification is higher | Higher; adults may voluntarily accept greater uncertainty | Pediatric decisions are made on behalf of the child, not by them |
| Marketing framing | Reassurance-based: immunity, growth, brain development | Enhancement-based: energy, performance, anti-aging | Marketing language in both categories often exceeds what evidence supports |
This table reflects general principles, not absolute rules. Individual needs, age, diet, and medical guidance still matter.

1. Differences in Physiological Scale and Metabolism
Children differ from adults in body mass, metabolic rate, organ maturity, and nutrient storage capacity. These differences influence how nutrients are absorbed, distributed, metabolized, and excreted.² A smaller body does not simply need a smaller version of the adult dose. The relationship is not proportional, and safety margins can narrow more quickly with ongoing use.
This is why pediatric dosing is typically conservative and age-stratified, not simply a fraction of adult recommendations.
2. Differences in Developmental Sensitivity
Children undergo rapid growth and neurological development. Certain nutrients are critical during specific windows, but excess intake during those same windows can also carry greater implications.³
This creates a dual consideration that shapes pediatric guidance: adequacy matters, and so does avoiding unnecessary excess. Adult supplementation often focuses on doing more or enhancing performance. Pediatric guidance is more concerned with meeting needs without adding unnecessary exposure.
3. Differences in Evidence Base
A significant portion of supplement research is conducted in adults.⁴ Pediatric data is often limited in duration, narrower in scope, and ethically constrained. A study in adults may be useful background, but it does not automatically answer a pediatric question.⁵
The limits of extrapolating adult data are examined in more depth in How to Interpret Supplement Research Without Getting Misled.
4. Differences in Risk Tolerance
Adults can voluntarily accept tradeoffs for themselves. Pediatric decisions operate differently: the decision is being made on behalf of the child, which raises the threshold for justification and emphasizes precaution.⁶
This does not mean pediatric supplementation is discouraged. It means the reason for use needs to be clearer, the dose more carefully considered, and the duration more intentional.
5. Differences in Duration Expectations
Adults often adopt long-term supplement routines. A supplement that becomes part of an adult wellness routine should not automatically become a child's long-term routine. In children, prolonged use is approached more cautiously because cumulative exposure matters more as the child grows and their needs change.⁷
Pediatric supplementation benefits from periodic reassessment, a principle explored further in What Safety Data Exists for Long-Term Supplement Use in Children?
Duration is part of the safety equation, not an afterthought.
6. Differences in Dosing Philosophy
Adult supplements frequently emphasize potency and higher intake. Pediatric guidance typically emphasizes meeting established requirements, avoiding excessive intake, and minimizing unnecessary complexity.⁹ The shift is from "more support" to "appropriate support."
This is examined in detail in When "More Is Better" Becomes Risky in Supplement Use.⁸
7. Differences in Marketing Framing
Children's supplements are often marketed through reassurance language: "immune support," "brain development," "growth support." Adult supplements tend to use enhancement language: "energy," "performance," "anti-aging." The words change by audience, but the same underlying problem can remain: marketing can make a product sound more specific or more proven than it is.¹⁰
For a practical way to read label language more critically, see How to Read a Supplement Label (Without Getting Misled).
A Practical Way to Apply This Distinction
Rather than applying adult supplementation habits to children, families can ask: Is this practice based on pediatric guidance or adult wellness trends? Does the dose reflect age-specific recommendations? Is this short-term support or indefinite use? Is there a clear reason?
This reframing keeps the focus on the child's age, need, dose, and duration, rather than adult supplement norms. The Supplement Decision Flow and Frameworks & Decision Tools Hub are both built around this kind of age- and need-specific thinking.
The Evidence-Informed Family Supplement Guide is coming soon. It will walk through children's multivitamins, probiotics, magnesium, dosing, safety tradeoffs, label review, and reusable worksheets for evaluating family supplement decisions with more clarity.
See What's Coming →Use the checklist to review ingredient identity, dosing, formulation quality, and supplement claims before relying on label language for pediatric decisions.
View the Checklist →Why This Matters for Families
Parents often encounter supplement advice in spaces that are not clearly pediatric: social media, adult wellness podcasts, product reviews, family routines, and general supplement labels. Without clear distinctions, adult habits can look like reasonable shortcuts for children. A supplement that is well-suited for an adult, at an adult dose, for an adult reason, may not be the right fit for a child at a different age, with different needs and a different safety margin.
A pediatric lens helps families avoid overgeneralizing while still leaving room for targeted support when there is a defined need. The goal is not to be more restrictive. It is to be more specific.
Pulling It All Together
Pediatric supplement decisions differ from adult supplement habits in physiology, evidence base, safety thresholds, risk tolerance, and duration. Children are not smaller versions of adult consumers, and their supplement decisions should not be adult routines scaled down.
Children's supplement decisions are strongest when they start with the child: age, diet, dose, duration, evidence, and the reason for use. The most useful question is not "What do adults take?" but: What is appropriate for this child, at this age, for this reason, and for this duration?
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- American Academy of Pediatrics. Pediatric Nutrition Handbook.
- Kearns GL et al. Developmental Pharmacology — Drug Disposition, Action, and Therapy in Infants and Children. New England Journal of Medicine.
- Georgieff MK. Nutrition and the Developing Brain: Nutrient Priorities and Measurement. American Journal of Clinical Nutrition.
- U.S. Department of Health and Human Services. Ethical Considerations in Pediatric Clinical Research.
- Ioannidis JPA. Why Most Clinical Research Is Not Useful. PLOS Medicine.
- American Academy of Pediatrics Committee on Bioethics. Ethical Conduct of Clinical Research in Children.
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes: Tolerable Upper Intake Levels.
- Institute of Medicine. Dietary Reference Intakes: Applications in Dietary Assessment.
- National Institutes of Health, Office of Dietary Supplements. Dietary Supplement Fact Sheets.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know.
All sources are freely accessible or summarized via NIH, FDA, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are included as examples based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
When “More Is Better” Becomes Risky in Supplement Use
It’s easy to assume that higher doses lead to better outcomes. But in supplementation, exceeding evidence-based ranges can narrow safety margins and increase unintended risk. This article explores where “more” stops helping — and starts working against you.
A clear explanation of how excessive dosing can introduce risk, and where upper limits begin to matter.
A label says "extra strength," "high potency," or "maximum support." You are comparing a standard dose to a stronger version and wondering whether the higher option offers more protection. A child is already taking a multivitamin, getting nutrients from fortified foods, and maybe a gummy on top of that. Choosing the stronger product can feel like covering all the bases.
That instinct is understandable. But with nutrients, more only helps up to a point. Once adequacy is reached, the question changes from whether more will help to whether more adds unnecessary exposure.
This article explores when increased intake may be justified, when it becomes unnecessary, and how to think about dose, safety margins, and cumulative exposure in a more balanced way.
The goal is proportionality, not fear.
Why "More Is Better" Feels Intuitive
The logic can feel reasonable: if too little is harmful, more must be safer. Supplements are associated with health support, deficiency is framed as consequential, and labels often emphasize potency as a mark of quality. Online advice can blur the line between meeting needs and doing more.
The problem is that nutrition does not always work that way. Higher doses can feel like greater protection without adding meaningful benefit once needs are already met. Understanding where that assumption breaks down is the more useful starting point.
How This Fits Within an Evidence-First Approach
Across this site, supplements are evaluated as tools designed to meet specific needs, not as performance enhancers. The useful question here is not "Which option is strongest?" It is "What dose fits the need without adding unnecessary exposure?"
This article uses the same evidence-first lens to examine dosing decisions, focusing on adequacy, safety thresholds, and current need rather than defaulting to higher intake. For a broader look at when supplementation may be worth considering at all, see How to Decide Whether a Supplement Is Worth Trying at All and Do Children Actually Need Supplements?
When Adequacy Is the Goal
Nutrient recommendations are generally designed to achieve adequacy, the level sufficient to prevent deficiency in most people. Moving beyond adequacy does not automatically produce additional benefit. In many cases, benefits plateau once sufficiency is reached, excess intake is excreted or accumulates depending on the nutrient, and risk begins to increase before additional benefit does.²
This is especially relevant in children, where margins between adequate and excessive intake may be narrower than in adults.
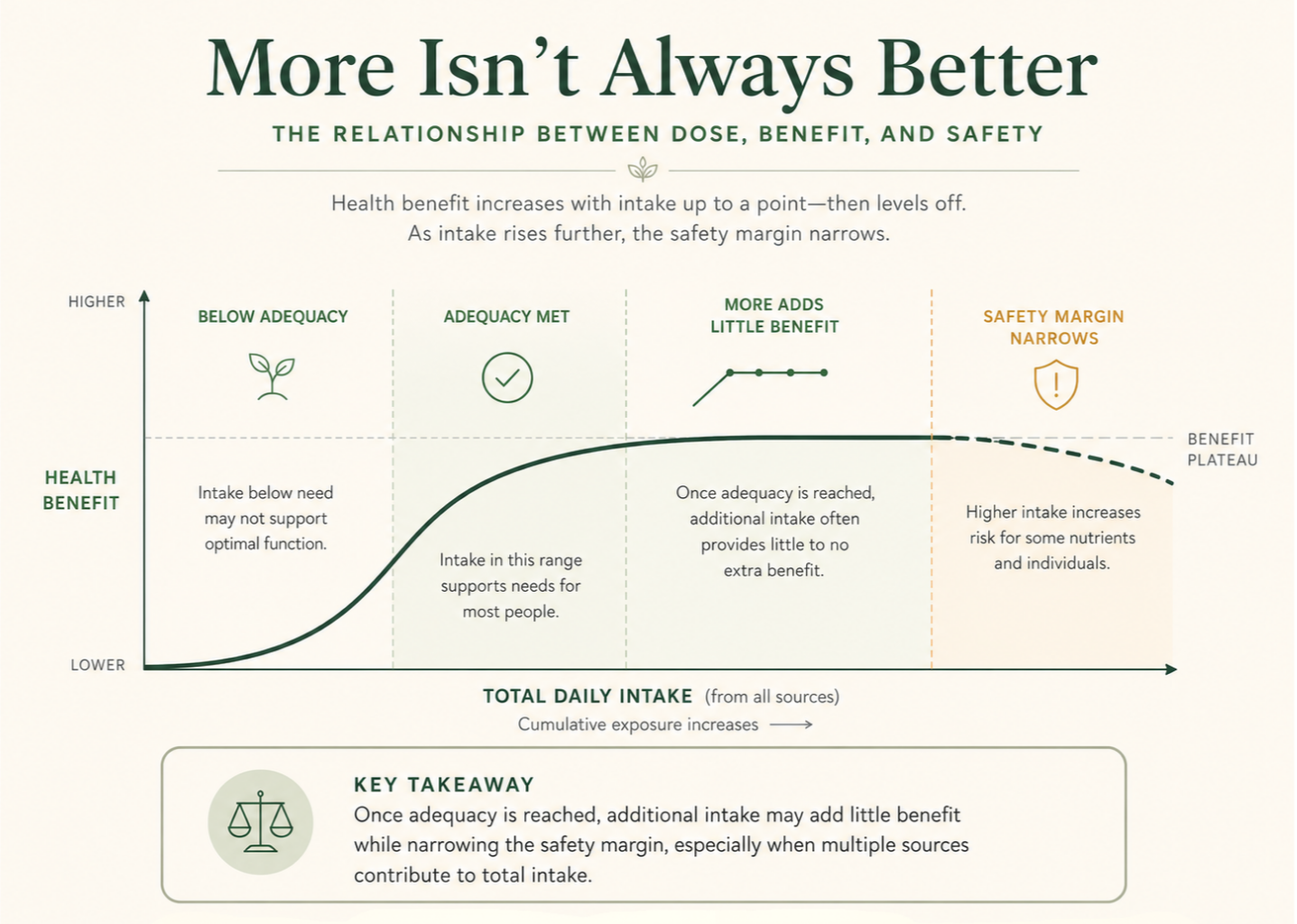
Upper Intake Levels and Safety Margins
For many nutrients, regulatory and scientific bodies establish tolerable upper intake levels (ULs).³ These levels are not ideal targets. They are thresholds beyond which the risk of adverse effects may increase.⁴ A number below the upper limit does not automatically mean the dose is useful, and a number near the upper limit should not be treated as a goal.
Long-term intake near or above these thresholds, particularly when multiple products are combined, can unintentionally reduce safety margins. Understanding that ULs are safety boundaries helps reframe what "high dose" means in practice.
Children Are Not Small Adults
Children differ from adults in body mass, metabolic rate, developmental stage, and nutrient requirements, all of which influence how nutrients are processed and tolerated.⁵ Higher doses that may be tolerated in adults do not automatically translate to pediatric safety.
This is explored in more depth in the broader discussion of pediatric versus adult supplement guidance, which covers why age-specific dosing and evidence matter independently of what works in adult populations.
When Higher Doses May Be Appropriate
There are situations where higher-than-baseline doses are used intentionally, including medically identified deficiencies, therapeutic short-term use, and specific clinical guidance.⁶ In these cases, increased intake is typically time-limited, monitored, and based on identified need. The key difference is intentionality, defined need, and duration, not potency alone.
Cumulative Exposure Is Often Overlooked
A child may not be taking a "high dose" from any single product. But the total can change quickly when a multivitamin, a gummy, fortified foods, and a single-nutrient supplement are all part of the routine. Each source contributes to total daily intake, and without periodic reassessment, cumulative intake can drift upward without anyone noticing.⁷
Form and dose tend to matter more than ingredient count alone, particularly when intake adds up across products. How to Read a Supplement Label (Without Getting Misled) explains how to identify overlapping nutrients across the products a family is already using.
Marketing and the Language of Potency
Labels often use "extra strength," "high potency," or "maximum support" as shorthand for a product being better or more protective. These phrases imply greater benefit but rarely clarify whether higher dosing is necessary or appropriate for the situation. Potency can sound like value, but value depends on whether the dose fits the need.
The Plateau Effect
A nutrient can be essential and still have a point where more stops helping. In nutritional science, many nutrients demonstrate a plateau effect: once physiological needs are met, additional intake does not improve outcomes.⁸ Continuing to increase intake beyond this plateau may add cost and complexity, reduce safety margins over time, and create a false sense of security.
For some nutrients, particularly those that accumulate in the body or come from multiple overlapping sources, higher intake can gradually narrow the gap between adequacy and excess. Recognizing this pattern helps counteract the assumption that a higher dose is a safer dose.
When More Stops Adding Value
| Dose Range | What It May Mean | Decision Lens |
|---|---|---|
| Below adequacy | Intake may not be sufficient to meet physiological needs | Evaluate dietary sources first; targeted supplementation may be appropriate |
| At adequacy | Needs are likely met; additional intake is unlikely to add meaningful benefit | Maintain without increasing; reassess if diet or situation changes |
| Above adequacy | Intake exceeds typical needs; benefit plateau has likely been reached | Question whether higher dose is necessary; monitor for cumulative exposure |
| Approaching upper limits | Safety margin is narrowing; especially relevant with combined supplement use | Review all sources of intake; dose reduction or product consolidation may be warranted |
| High-dose use | Risk of adverse effects may increase; particularly relevant in children and long-term use | Use only with clear clinical reason, defined duration, and professional oversight |
Increasing dose adds value only up to adequacy. Beyond that, safety margins may narrow.
A Practical Way to Evaluate Dose
Instead of asking "Is this the strongest option?" it can help to ask: What is the identified need? Is current intake already adequate? Does increasing the dose meaningfully reduce risk? Is higher dosing temporary or indefinite?
This reframing keeps the focus on whether the dose is useful, necessary, and proportionate. The Frameworks & Decision Tools Hub includes structured tools that apply this same thinking across different supplement categories.
The Evidence-Informed Family Supplement Guide is coming soon. It will walk through dosing, label review, cumulative exposure, safety margins, and category-specific tradeoffs for evaluating family supplement decisions with more clarity.
See What's Coming →Preventing Dose Drift
Dose drift happens when a supplement is increased or layered on out of caution, but the reason for the higher dose is never clearly defined. The starting point makes sense, the habit continues, and the dose becomes part of the routine without periodic review. The Supplement Decision Flow walks through adequacy, safety margins, cumulative exposure, and duration before adjusting dose upward.
Use the decision flow to work through adequacy, safety margins, cumulative exposure, and duration before adjusting dose upward. No rankings. No performance framing.
Use the Supplement Decision Flow →Why This Matters for Families
Parents often choose higher-dose products from a place of care. It can feel responsible to pick the stronger option, especially when labels suggest it provides more support. But if a child's needs are already met, a higher dose may not add more protection. It may simply add more exposure, especially when products are being combined.
Understanding where benefit plateaus and where safety margins narrow supports calmer, more proportionate decisions. The instinct to cover all bases is understandable. The goal is to make sure "covering all bases" means meeting the need, not exceeding it.
Pulling It All Together
Once adequacy is achieved, increasing intake does not guarantee additional benefit and may reduce safety margins over time. An evidence-informed approach prioritizes adequacy over potency, defined need over habit, and periodic review over automatic continuation.
A supplement dose is most useful when it addresses a defined need without adding unnecessary exposure. Once needs are met, the strongest option is not automatically the best-matched one. The most useful question is not "Is this the strongest option?" but whether this dose meaningfully addresses the need, or simply increases exposure.
Use the checklist to review ingredient identity, dosing, formulation quality, and supplement claims before deciding whether a product and dose fit the need.
View the Checklist →Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- Heaney RP. Nutrient Dose-Response Relationships and the Plateau Effect. American Journal of Clinical Nutrition.
- Institute of Medicine (National Academies). Dietary Reference Intakes: Applications in Dietary Assessment.
- National Academies of Sciences, Engineering, and Medicine. Tolerable Upper Intake Levels for Vitamins and Minerals.
- National Academies of Sciences, Engineering, and Medicine. Risk Assessment Model for Establishing ULs.
- National Institutes of Health. Pediatric Research and Age-Specific Dosing Principles.
- National Institutes of Health, Office of Dietary Supplements. Iron, Vitamin D, and Fat-Soluble Vitamin Fact Sheets.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know About Safety.
- Hathcock JN. Risk Assessment for Vitamins and Minerals. American Journal of Clinical Nutrition.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
What Safety Data Exists for Long-Term Supplement Use in Children?
Many supplements are studied over weeks or months — but what do we actually know about long-term use in children? This article reviews the available safety data, highlights evidence gaps, and explains how to interpret duration and risk over time.
A focused look at the available safety data, and where meaningful gaps still exist.
A supplement often starts with a clear reason: picky eating, a vitamin D concern, a probiotic after antibiotics, a multivitamin during a difficult eating phase. Months later, it may still be part of the daily routine. The original reason made sense, nothing obvious went wrong, and the habit continued.
But tolerating a supplement this week is a different question from knowing whether continued use still makes sense after months or years. Many children take supplements daily for extended periods, yet the evidence base for long-term use is often assumed rather than clearly understood.
This article examines what is known, and what remains uncertain, about the long-term safety of supplement use in children, using an evidence-informed, risk-based approach. Rather than offering reassurances or alarm, it focuses on how safety data is generated, where limitations exist, and how families can think about duration and exposure thoughtfully.
The goal is informed caution, not fear.
Why Long-Term Safety Is a Different Question
Short-term tolerance does not automatically predict long-term appropriateness.¹ A supplement that causes no immediate side effects may still deserve reassessment when used daily, over extended periods, or during critical stages of growth and development. A product can be tolerated and still warrant a second look if the original reason for use has changed.
Long-term exposure introduces questions around accumulation, dose appropriateness over time, and shifting physiological needs.² Understanding this distinction helps explain why long-term pediatric safety data is often more limited than the familiarity of a product might suggest.
Tolerated today does not automatically mean appropriate indefinitely.
Long-term use introduces different questions: cumulative exposure, changing needs, dose over time, and whether the original reason for supplementation still applies.
How This Fits Within an Evidence-First Approach
Evaluating long-term safety requires careful attention to what evidence exists and what does not. The useful question is not "Is this safe forever?" It is "What do we know, what remains uncertain, and does continued use still make sense for this child at this dose?"
This article uses the same evidence-first lens applied throughout this site to examine pediatric supplement safety, with particular focus on duration, exposure, and uncertainty.
Long-term safety is not answered by reassurance alone.
The goal is not to assume harm or safety. It is to understand what evidence exists, what remains uncertain, and how duration, dose, and the child's current needs change the decision.
What "Safety Data" Typically Means in Pediatrics
Families may expect safety data to be one clear answer. In reality, it is often pieced together from different types of evidence.³ In pediatric contexts, safety data typically comes from a mix of sources rather than long-term randomized trials, including short-term clinical studies, observational data, post-market surveillance, and extrapolation from adult data. Each of these contributes information, but none alone provides a complete picture of long-term safety.
Understanding how safety evidence is assembled helps set realistic expectations about what can and cannot be concluded from it.
Sources of Pediatric Supplement Safety Data, and Their Limits
| Evidence Source | What It Can Tell Us | Key Limitations | How to Interpret It |
|---|---|---|---|
| Short-term clinical studies | Tolerance, short-term side effects, early safety signals | Often limited duration, small sample sizes, and narrow populations | Useful for short-term safety, but not enough to assume long-term safety |
| Observational data | Real-world patterns of use and potential associations | Can be affected by confounding factors, inconsistent reporting, and unclear exposure details | Helpful for signals, but rarely definitive on its own |
| Post-market surveillance | Reported adverse events after products are available | Underreporting, incomplete context, and difficulty proving cause | Important for safety signals, but not a full safety profile |
| Adult data extrapolation | General physiology, dose considerations, and known risks in adults | Children are not simply smaller adults; growth and development change the picture | Useful background, but pediatric relevance must be interpreted carefully |
| Upper intake levels and nutrient risk frameworks | Reference points for avoiding excessive intake | Often nutrient-specific and not product-specific | Helpful for dose boundaries, especially with repeated or combined exposure |
| Historical use | Patterns of long-standing use and broad familiarity | Familiar use is not the same as systematic long-term safety evidence | Contextual only; should not replace dose, duration, and risk evaluation |
Long-term pediatric safety data usually comes from multiple evidence sources. Each source can be useful, but none should be treated as a complete answer on its own.
Safety evidence is often pieced together, not handed to families as one clear answer.
That does not mean families should panic. It means long-term decisions deserve more attention to duration, dose, individual need, and periodic reassessment.
Why Long-Term Pediatric Trials Are Rare
There are practical and ethical reasons why long-term supplement trials in children are uncommon.⁴ Challenges include ethical constraints around prolonged exposure, difficulty controlling diet and environment over time, limited funding for non-pharmaceutical interventions, and rapidly changing nutritional needs during growth.
This does not mean no one cares about long-term safety. It means these studies are genuinely difficult to conduct well. The absence of long-term data often reflects structural limitations in how research is resourced and designed, not negligence.
A lack of long-term trials does not automatically mean danger, but it does limit certainty.
For families, that uncertainty should lead to proportionate caution: define the reason for use, avoid unnecessary exposure, and revisit the decision as needs change.
What Can Be Inferred, and What Cannot
In some cases, long-term safety is inferred from known nutrient physiology, established upper intake levels, and patterns of historical use. These inferences can be useful, but they are not equivalent to direct evidence.⁵ Useful inference is not the same as a complete answer.
Evidence is typically tied to specific compounds, doses, and contexts, not to supplement categories as a whole. Understanding this distinction helps prevent overconfidence in generalized safety claims.
What Can Be Inferred, and What Still Needs Caution
Dose, Duration, and Accumulation Matter
Long-term safety is rarely about whether a nutrient is "safe" in isolation. The question changes when a supplement becomes part of the daily background. Cumulative exposure, dose relative to need, duration of use, and changes in diet over time all shape how appropriate continued use remains.
Form and dose tend to matter more than ingredient count alone, especially with ongoing use.⁶ A dose appropriate for short-term support may not remain appropriate indefinitely, particularly as a child's diet, weight, and nutritional needs evolve.
Children's Needs Change Over Time
Children do not stay in the same nutritional situation for long.⁷ Growth, development, and dietary patterns shift continuously. A supplement that made sense during a season of picky eating, low intake, or recovery may not need to remain in the routine indefinitely. What was reasonable at one stage may become unnecessary, or even excessive, later.
This is why long-term supplement decisions benefit from periodic reassessment rather than automatic continuation.
For ongoing supplement use, reassess periodically:
Common Assumptions About Long-Term Use
Several assumptions often appear in discussions of long-term supplementation: "It's just a vitamin, so it's harmless." "More coverage is better than less." "If it hasn't caused problems yet, it won't."
These assumptions are understandable. Supplements often feel familiar and low-risk, and that familiarity can make continued use feel like a neutral choice. But familiar does not mean there is nothing to evaluate. These assumptions can quietly obscure more relevant questions about necessity, dose, and duration.⁸
Familiar does not automatically mean risk-free.
The most relevant safety questions are not whether a supplement feels common or gentle. They are whether the dose, duration, and ongoing need still make sense.
Risk Management, Not Zero Risk
No intervention, dietary or otherwise, is entirely risk-free. The goal is not to eliminate every possible risk. It is to make the decision proportional to the need. Long-term safety decisions involve weighing potential benefit, potential risk, likelihood of deficiency, and the availability of alternative approaches.⁹
This framing aligns with how safety decisions are made in other areas of child health, where the aim is proportional, thoughtful evaluation rather than certainty.
A Practical Way to Think About Duration
Rather than viewing supplementation as an open-ended decision, it helps to ask: What prompted supplementation initially? Has that reason changed? Is continued use still justified? Should dose or form be revisited?
Duration should be part of the decision, not something that disappears once the supplement becomes routine. A time-bounded approach supports more intentional use and makes it easier to adjust as the child's needs evolve.
Long-term supplementation should be revisited, not put on autopilot.
A supplement that made sense at one point may not need to continue indefinitely. A time-bounded review helps families make changes without guilt, fear, or unnecessary complexity.
Using a Structured Decision Tool
Long-term supplementation decisions benefit from clarity around purpose, duration, and reassessment. A structured decision flow can help think through need, exposure, and proportional risk, especially when use extends beyond short-term support.
Use the decision flow to review need, exposure, duration, and proportional risk when supplement use extends beyond short-term support.
Use the Supplement Decision Flow →Use the checklist to review ingredient identity, dose, formulation quality, and evidence signals before relying on supplement claims or continuing routine use.
View the Checklist →Why This Matters for Families
Families often continue supplements because the original reason made sense, the product is already in the routine, and nothing obvious has gone wrong. That is understandable. Starting a supplement for a defined reason and tolerating it without issue does not mean the original reason is still present.
Children's diets, growth patterns, and needs change. Revisiting long-term supplement use helps families make adjustments without fear, guilt, or the feeling that they did something wrong. The question is not whether starting the supplement was a mistake. The question is whether continuing it still fits the current situation.
Explore structured EFW resources for evaluating supplement tradeoffs, including dose, duration, exposure, formulation quality, and real-world use.
Explore the Frameworks Hub →Pulling It All Together
Long-term supplement use in children is an area where certainty is often assumed but evidence is more nuanced. An evidence-informed approach acknowledges those limits, prioritizes dose and duration, and treats supplementation as a revisitable decision, not a permanent one.
A supplement is easiest to keep when it becomes part of the routine. That is exactly why long-term use deserves a periodic pause. The most useful question is not "Is this supplement safe forever?" It is whether continued use meaningfully reduces risk in this child, at this stage, and at this dose.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- Ioannidis JPA. Why Most Clinical Research Is Not Useful. PLOS Medicine.
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes: Tolerable Upper Intake Levels.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know About Safety.
- U.S. Department of Health & Human Services. Ethical Considerations in Pediatric Research.
- National Institutes of Health, Office of Dietary Supplements. Dietary Supplement Fact Sheets (Safety and Physiology Sections).
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes: Risk Assessment Framework.
- Centers for Disease Control and Prevention. Nutrition, Growth, and Development Guidance.
- National Institutes of Health, Office of Dietary Supplements. Fat-Soluble Vitamins Fact Sheets (Vitamin A, D, E, K).
- National Academies of Sciences, Engineering, and Medicine. Framework for Assessing Nutrient Risk and Benefit.
All sources are freely accessible via NIH, FDA, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Are Gummy Vitamins Worth It? An Evidence-Informed Look at Tradeoffs
An evidence-informed look at the tradeoffs of gummy vitamins, including formulation limitations, dosing constraints, and when gummies may—or may not—make sense for families.
A structured evaluation of gummy vitamins, focusing on tradeoffs in dosing, stability, and formulation quality.
Your child will not take a capsule. Powders go untouched. A chewable tablet was rejected after one try. Gummies, though, get taken without a fight. That matters, and it is a legitimate reason to consider the format.
Gummy vitamins are one of the most popular supplement formats for families precisely because they solve a real practical problem. But the format that makes a supplement easier to take also changes what the supplement can contain, how precisely it can be dosed, what sweeteners are involved, and how stable the nutrients remain over time.
This article examines gummy vitamins with that in mind. Rather than asking whether gummies are "good" or "bad," it focuses on what changes when vitamins are delivered in gummy form, what tradeoffs are introduced, and when this format may or may not make sense for families.
Why Gummy Vitamins Became So Common
Gummy vitamins became common because they solve a practical problem: acceptance. ¹ For many children, swallowing capsules or tolerating powders is difficult. Gummies offer improved acceptance, simpler daily routines, and a format that is much easier to build into consistent use.
These advantages explain their popularity. They do not eliminate the need to evaluate what else the format changes.
What Gummy Vitamins Can Help With, and What They Do Not Solve
How This Fits Within an Evidence-First Approach
When evaluating supplements, delivery format is one variable among many. The same core questions apply regardless of whether a vitamin is delivered as a gummy, capsule, powder, or liquid: Is the dose meaningful? Is the formula appropriate? Are the tradeoffs acceptable for daily use?
A gummy may make a supplement easier to take. It does not answer those questions on its own.
What Changes When Vitamins Are Made Into Gummies
Gummy vitamins are not simply capsules in a different shape. ² The gummy format changes what the manufacturer has to solve for. A product must maintain the right texture and stability, manage moisture content, incorporate flavoring and sweeteners, and meet shelf-life requirements.
These requirements influence formula decisions in ways that are often invisible on the label. What fits in a capsule may not fit in a gummy, and what is stable in a dry tablet may behave differently in a moist, chewy format.

Dosing Precision Is Often Reduced
Unlike capsules or tablets, gummies typically contain lower amounts of individual nutrients per unit. ³ This is partly a space constraint and partly a taste and stability constraint. The result is that precise dosing can be more difficult, especially when children fall between age-based dose ranges, only a partial dose is appropriate, or multiple nutrients are combined in one product.³
A gummy can be easier to take, but the amount per serving still needs to match the reason the supplement is being used. Convenience matters. So does whether the dose is meaningful for the stated purpose.
Convenience is useful only if the dose still fits the purpose.
A gummy format may make a supplement easier to take, but the amount per serving still needs to be meaningful, realistic, and appropriate for the person using it.
Sugar, Sweeteners, and Functional Tradeoffs
To be palatable, gummy vitamins require sweeteners.⁴ These may include sugars, sugar alcohols, or non-nutritive sweeteners, each with different implications for dental health, gastrointestinal tolerance, and daily exposure over time.
The presence of sweeteners does not automatically disqualify a product. It is a tradeoff that deserves consideration, particularly for daily use.⁴
Before choosing a gummy vitamin for daily use, ask:
Stability and Shelf-Life Considerations
Some vitamins are sensitive to heat, light, and moisture. Because gummies contain water and require texture, they create a different stability environment than dry formats like capsules or tablets. Manufacturers must balance vitamin stability against taste, texture, and packaging limitations.⁵
This can affect which forms of vitamins are used and how well potency is maintained over the product's shelf life. It is not a reason to dismiss gummies, but it is a reason to understand that delivery format affects more than appearance.⁶
A gummy is not just a different shape. It is a different formulation environment.
Moisture, texture, sweeteners, and shelf-life constraints can influence which nutrients are included, which forms are used, and how much can realistically fit.
Where Gummy Vitamins Are Most Commonly Used
Gummy formats appear most often in children's multivitamins, vitamin D, vitamin C, and general daily wellness products. These categories depend heavily on routine use and acceptance, which makes gummies a natural fit from a practical standpoint.
In these categories, marketing often emphasizes natural flavors, colors, or sourcing, sometimes in ways that shift attention away from what matters more: whether the dose is meaningful, whether the formula fits the intended use, and whether daily sweetener exposure is worth considering.⁷
A one-page reference for reviewing ingredient identity, dose, form, formulation quality, and supplement claims more clearly.
View Checklist →Gummies vs. Other Delivery Formats
Compared with capsules, powders, or liquids, gummy vitamins tend to prioritize ease of use over formula flexibility. Other formats may offer more precise dosing, fewer formulation constraints, and broader options for nutrients that are harder to stabilize in gummy form.⁸
This does not make gummies the wrong choice. It does mean they serve a different primary purpose and should be evaluated with that in mind.
Gummy Vitamins Compared With Other Delivery Formats
| Format | Common Strengths | Common Tradeoffs | Best Fit |
|---|---|---|---|
| Gummies | Palatable; easy to build into routines; helpful for children who resist pills or powders | Lower dosing flexibility; sweeteners; stability constraints; limited formulation space | When acceptance and routine consistency are the main barriers |
| Capsules or tablets | More formulation flexibility; often easier to dose precisely; fewer taste constraints | Harder for some children or adults to swallow; less appealing for routine use | When dosing precision, ingredient density, or fewer sweeteners matter more |
| Powders | Flexible serving size; can allow higher amounts; easy to mix into foods or drinks | Taste, texture, measuring accuracy, and consistency can be issues | When flexible dosing is useful and the user tolerates mixing |
| Liquids | Adjustable dosing; easier for some children; useful when swallowing is difficult | Stability, taste, preservatives, and measuring accuracy may matter | When dose adjustment or swallowing difficulty is the primary concern |
Explore structured EFW resources for evaluating supplement tradeoffs, including delivery format, dosing precision, formulation flexibility, tolerance, and long-term use.
Explore the Frameworks Hub →Managing Expectations Around Gummy Vitamins
Gummies can make a routine easier, and that is not a small thing. A supplement that gets taken is more useful than one that sits in a cabinet. But ease can also make a supplement feel like a harmless daily habit, even when the dose, sweeteners, overlap with other products, or long-term need still deserve attention.⁹
An evidence-first approach treats gummies as one option among several, appropriate in some situations and less suitable in others, depending on the specific child, dose, and goal.¹
A gummy vitamin is a format choice, not a quality guarantee.
For families, the goal is not to avoid or prefer gummies automatically. The goal is to understand whether the format supports the actual need without hiding important tradeoffs around dose, stability, sweeteners, or long-term use.
Why This Matters for Families
For many families, gummies feel like a practical compromise. If a child will only take a gummy, that is a real consideration and it matters. But the decision should still include whether the supplement is needed, whether the dose is meaningful, whether the sweeteners are reasonable for daily use, and whether another format would better fit the goal.
Gummies are not a shortcut to quality, and they are not automatically a second-best option. Understanding what the format does and does not change helps shift the decision away from convenience or marketing and toward what matters for the situation.
Pulling It All Together
Gummy vitamins are not inherently inferior or inherently superior to other supplement formats. They represent a shift in priorities.
When vitamins are delivered as gummies, acceptance and palatability are often improved. In exchange, formula flexibility, dosing precision, ingredient simplicity, and stability may be constrained. Understanding those tradeoffs allows families to make decisions based on context rather than format preference.
A gummy vitamin is most useful when the format solves a real use problem and the dose, formula, and tradeoffs still fit the reason for taking it. In some situations, improved acceptance outweighs the constraints. In others, precision or long-term stability matters more. An evidence-informed approach does not rank delivery formats. It asks whether the chosen format aligns with the intended purpose, dosing needs, and duration of use.
Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →If you're weighing a specific gummy vitamin or ingredient and aren't sure how it fits the tradeoffs discussed here, you can submit a question to help guide future Evidence First Wellness content. This is not medical advice, and I cannot evaluate individual health conditions, but reader questions help shape future explanations about evidence, quality considerations, and common marketing claims.
Submit a Question →References and Further Reading
- National Institutes of Health Office of Dietary Supplements. Consumer Guidance on Dietary Supplements.
- Harvard T.H. Chan School of Public Health. Vitamins and Supplements: Understanding Different Forms.
- National Institutes of Health Office of Dietary Supplements. Multivitamin/Mineral Supplements Fact Sheet.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know (Sweeteners & Labeling).
- National Institutes of Health Office of Dietary Supplements. Stability of Vitamins and Minerals in Supplements.
- Allen LV. Stability of oral formulations. International Journal of Pharmaceutical Compounding.
- U.S. Food & Drug Administration. Dietary Supplement Labeling Guide.
- American Dental Association. Sugar and Children's Oral Health.
- National Institutes of Health Office of Dietary Supplements. Dietary Supplement Fact Sheets (Formulation & Stability).
All sources are freely accessible or summarized via NIH, FDA, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are discussed based on formulation characteristics, not sponsorship, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Magnesium for Families: An Evidence-Informed Guide to Forms, Dosing, and Tradeoffs
An evidence-informed guide to evaluating magnesium for families, explaining how different forms, dosing ranges, and tradeoffs affect use and why simple ingredient lists rarely tell the full story.
A clear evidence-informed guide to magnesium forms, dosing considerations, and when use may be appropriate.
Magnesium is one of the most frequently discussed minerals in family wellness, and one of the most misunderstood.
You may be seeing it connected to sleep, calm, stress, digestion, or relaxation. A label promises "gentle absorption" or "relaxing magnesium." A powder is marketed for bedtime routines. A gummy suggests it supports a calmer child. Parents trying to sort through these claims quickly encounter a category where the products look very different, the language sounds reassuring, and it is not always obvious what distinguishes one option from another.
Magnesium decisions are shaped less by the mineral itself and more by the form, dose, tolerance, total intake, and context. For families, those factors matter far more than marketing claims. This guide explains how to evaluate magnesium with that in mind, without assuming supplementation is always the right step.
Why Magnesium Comes Up So Often
Magnesium plays a role in hundreds of biochemical processes, including neuromuscular signaling, energy metabolism, and enzyme function.¹ Because of this broad involvement, magnesium is frequently mentioned in wellness conversations. But broad biological relevance does not automatically mean supplementation is useful or necessary. The mineral being important is not the same as a supplement being needed.¹
For families, magnesium often enters the conversation around picky eating or limited diet variety, growth and development phases, digestive tolerance, and sleep routines or transitions. These are reasonable circumstances. They are also circumstances where the right response depends heavily on what else the child is eating, what form is being considered, and what the actual goal is.
Understanding how magnesium is delivered matters just as much as whether it is present at all.
How This Fits Within an Evidence-First Approach
Magnesium is evaluated here using the same lens applied throughout this site: What form is being used? How much is provided? Who is it intended for? What are the tradeoffs?
Rather than asking whether magnesium is "good" or "bad," the more useful question is whether a specific magnesium product makes sense for the situation, dose, form, and child. That question requires looking past the label and into how the product is built.
Magnesium Is Not One Thing: Understanding Forms
Magnesium appears on labels as a single ingredient, but it is always delivered as a compound, bound to another molecule. That compound affects how well the mineral is absorbed, how well it is tolerated, and how practical it is to use.²

Magnesium Glycinate
Bound to the amino acid glycine, magnesium glycinate is generally well tolerated and less likely to cause digestive upset. It is often chosen when a gentler form is a priority. This does not make it a proven solution for sleep or calm, but its tolerability profile makes it a common choice when digestive sensitivity is a concern.
Magnesium Citrate
More readily absorbed, magnesium citrate draws water into the intestine. This can be useful in some circumstances, but may cause loose stools at higher intakes.³ It is not the right form for every child or every goal.
Magnesium Oxide
Magnesium oxide has high elemental magnesium content but lower absorption efficiency compared to other forms. It is common in lower-cost products.² That does not make it useless, but the difference in bioavailability is worth understanding when comparing products.
Other Forms (Malate, Threonate, etc.)
These appear in more specialized products with varying levels of evidence. They are less common in family-focused formulas and often included at lower doses. The research base for most of these forms in pediatric contexts is limited.
Why Form Matters for Families
For children, tolerance and consistency can matter more than theoretical absorption. A form that causes stomach upset, is difficult to mix, tastes unpleasant, or cannot be used consistently may not be a good fit, even if it looks favorable on paper. Choosing a form with strong absorption characteristics does not help if the child refuses it or tolerates it poorly.
Magnesium should always be considered alongside the delivery method, total daily intake, and dietary sources already present in the child's routine.
Magnesium Supplement Formats Families Commonly Encounter
Once form and dose are understood, magnesium supplements tend to fall into a few practical delivery categories. These categories are not specific brands, and are the most useful way to compare options, particularly for children.
Not all formats are equally available or appropriate in pediatric products. The examples below reflect what families commonly encounter in the current supplement market.
1. Powdered Magnesium (Drink Mixes)
Powdered magnesium products are among the most common magnesium-focused options formulated for children. They are often chosen for flexible dosing, use by older children or teens, and families who prefer to avoid chewables. Powders allow dose adjustments and make it easier to tailor intake based on tolerance. Taste and texture can limit acceptance, and some forms, such as magnesium citrate, may cause gastrointestinal discomfort at higher intakes.
One commonly encountered example is Natural Vitality Calm Kids Magnesium Powder.
2. Liquid Magnesium
Liquid magnesium products are sometimes considered for children because they allow small, incremental dosing. However, magnesium-only products specifically labeled for pediatric use are uncommon. Families more often encounter magnesium in liquids combined with other nutrients, such as calcium or vitamin D. These formulas require careful label review, since magnesium may not be the primary nutrient and total intake can be harder to track across multiple supplements.
As a result, liquid magnesium is best evaluated as a category rather than assumed to be a straightforward option.
3. Chewable or Gummy Magnesium
Chewable and gummy magnesium products marketed for children are typically combination formulas, not magnesium-only supplements. They are often chosen for taste acceptance, routine consistency, and children who are unlikely to take powders, capsules, or liquids. Because magnesium contributes bitterness and bulk, chewable products usually contain lower magnesium amounts alongside other nutrients or sweeteners. Evaluating total magnesium intake carefully matters here, especially when used alongside multivitamins or fortified foods. One commonly encountered example is Olly Kids Chillax.
4. Combination Magnesium Products
Some children's supplements include magnesium as part of broader formulas, such as bone or growth-focused products that combine calcium, vitamin D, and magnesium. These products are often chosen for convenience, but they introduce real tradeoffs: magnesium is not the primary focus, dosing flexibility is limited, and cumulative intake across supplements can be difficult to assess.
Combination products highlight why understanding stacking and total intake matters more than the presence of any single ingredient.
One commonly encountered example is ChildLife Essentials Liquid Calcium with Magnesium.
Product mentions in these categories are selected based on formulation characteristics, not sponsorship, and are intended for educational comparison only.
Use the checklist to compare magnesium forms, dosing, and formulation tradeoffs in a more structured way.
View the Checklist →Dosing: More Is Not Better
Magnesium has established upper intake limits for supplemental forms. Excess intake from supplements can lead to gastrointestinal upset, and at very high intakes, electrolyte imbalance becomes a concern.⁴ This is not a reason to avoid magnesium, but it is a reason to be deliberate about how much is coming from all sources.
For families, this means accounting for magnesium from food and supplements together, and paying attention to overlap across multivitamins, gummies, powders, and combination products.⁴ The total matters, not just the amount on any one label.
Food First, Supplement Second
Many foods contribute meaningful magnesium, including whole grains, legumes, nuts and seeds, and leafy greens. For children with reasonably varied diets, these sources may already be contributing significantly to daily needs, and supplementation may not be necessary.
For families navigating picky eating, this is worth knowing without being discouraging: even partial dietary exposure to magnesium-containing foods can be meaningful. This aligns with professional guidance that emphasizes food first and reserves supplementation for situations where intake, tolerance, or specific circumstances suggest it may be helpful.⁵
The Evidence-Informed Family Supplement Guide is coming soon. It will walk through magnesium, children's multivitamins, probiotics, label claims, dosing, safety tradeoffs, and reusable worksheets for making supplement decisions with more clarity.
See What's Coming →Why This Matters for Families
Magnesium is often added to routines with good intentions. Parents want to support sleep, calm, digestion, or general wellness, and magnesium appears in many products connected to all of those things. That is understandable. But without clarity around form, dose, total intake, and tolerance, it is easy to add a product that is poorly matched to the situation, redundant with what a child is already consuming, or difficult to evaluate over time.
For families, thoughtful evaluation helps prevent unnecessary supplementation and reduces the risk of unintended effects, especially when products are used consistently. Understanding what the supplement is providing, and whether it fits the actual need, is more useful than responding to a label claim.
Pulling It All Together
Magnesium decisions are rarely about finding a single best form. Different forms vary in absorption characteristics, gastrointestinal tolerance, dosing flexibility, and practicality. What works well for one child may not work for another, and what looks strong on paper may not be realistic in daily use.
The most useful approach is to clarify what is being addressed before comparing products. What dose aligns with age and need? How does the chosen form fit the child's tolerance and the household routine? Is supplementation necessary given the diet, or is it being added out of habit or precaution?
In many cases, differences between forms matter less than thoughtful dosing and realistic expectations. When magnesium is considered with that lens, the decision becomes less about finding the strongest option and more about deciding whether supplementation fits the actual need, dose, and context.
Magnesium does not need to be framed as essential for every family to be worth evaluating. In many cases, understanding when it may not be necessary is just as important as knowing when it might be considered.
Where Magnesium Fits in the Broader Supplement Conversation
If you are new to evaluating supplements, these foundational guides may help:
- How I Evaluate Supplements as a Biomedical Engineer and Quality Professional
- How to Read a Supplement Label (Without Getting Misled)
Magnesium decisions follow the same logic applied across vitamins, minerals, and probiotics: formulation quality, dose, and real-world use matter more than claims.
Browse side-by-side comparison content designed to help families evaluate supplement categories through formulation, dose, quality signals, and tradeoffs.
View All Comparisons →Get Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families better interpret supplement claims, research findings, and formulation tradeoffs.
Subscribe to the Digest →References and Further Reading
- National Institutes of Health Office of Dietary Supplements. Magnesium Fact Sheet for Health Professionals.
- Schuette SA et al. Bioavailability of magnesium diglycinate vs magnesium oxide. Journal of the American College of Nutrition.
- National Institutes of Health Office of Dietary Supplements. Magnesium Fact Sheet (Adverse Effects and Tolerance).
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes for Magnesium.
- Harvard T.H. Chan School of Public Health. Magnesium and Health.
All are publicly accessible via NIH, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase, at no additional cost to you. Products are referenced for educational comparison only, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
A clear evidence-informed guide to magnesium forms, dosing considerations, and when use may be appropriate.
Magnesium is one of the most frequently discussed minerals in family wellness — and one of the most misunderstood.
It's often framed as a solution for sleep, stress, digestion, or general "calm," but magnesium decisions are shaped less by the mineral itself and more by the form, dose, tolerance, and context. For families, those factors matter far more than marketing claims.
This guide explains how to evaluate magnesium for families using an evidence-informed framework — without assuming supplementation is always necessary.
Why Magnesium Comes Up So Often
Magnesium plays a role in hundreds of biochemical processes, including neuromuscular signaling and energy metabolism and enzyme function.¹ Because of this broad involvement, magnesium is frequently mentioned in wellness conversations. That breadth also makes it easy to overgeneralize its benefits or assume supplementation is universally helpful.¹
For families, magnesium often enters the conversation when navigating selective eating, growth and development phases, digestive tolerance, and sleep routines or transitions.
Understanding how magnesium is delivered matters just as much as whether it's present at all.
How This Fits Within an Evidence-First Framework
Magnesium is evaluated using the same framework applied throughout this site: What form is being used? How much is provided? Who is it intended for? What are the tradeoffs?
Rather than asking whether magnesium is "good" or "bad," the more useful question is whether a specific formulation makes sense for a specific situation.
Magnesium Is Not One Thing: Understanding Forms
Magnesium must be bound to another compound, and that binding affects absorption and tolerance.²

Magnesium Glycinate
Bound to the amino acid glycine, magnesium glycinate is generally well tolerated and less likely to cause digestive upset. It is often prioritized when gentler absorption is desired.
Magnesium Citrate
More readily absorbed, magnesium citrate draws water into the intestine. This can be useful in some contexts, but may cause loose stools at higher intakes.³
Magnesium Oxide
Magnesium oxide has high elemental magnesium content but lower absorption efficiency. It is common in lower-cost products, but less bioavailable than other forms.²
Other Forms (Malate, Threonate, etc.)
These appear in more specialized formulations with varying levels of evidence, and are often included at lower doses. They appear less frequently in family-focused products.
Why Form Matters for Families
For children, tolerance and consistency matter more than theoretical absorption. A form that causes digestive discomfort or is difficult to administer consistently may not be appropriate — even if it looks favorable on paper.
Magnesium should always be considered alongside delivery method, total daily intake, and dietary sources already present.
Magnesium Supplement Formats Families Commonly Encounter
Once form and dose are understood, magnesium supplements tend to fall into a few practical delivery categories. These categories — not specific brands — are the most useful way to compare options, particularly for children.
Importantly, not all formats are equally available or appropriate in pediatric products. The examples below reflect what families commonly encounter in the current supplement market.
1. Powdered Magnesium (Drink Mixes)
Powdered magnesium products are among the most common magnesium-forward options formulated for children. They are often prioritized for flexible dosing, older children or teens, and families avoiding chewables. Powders allow dose adjustments and make it easier to tailor intake based on tolerance. However, taste and texture can limit acceptance, and some forms — such as magnesium citrate — may cause gastrointestinal discomfort at higher intakes.
One commonly encountered example is Natural Vitality Calm Kids Magnesium Powder.
2. Liquid Magnesium
Liquid magnesium products are sometimes discussed for children because they allow small, incremental dosing. However, magnesium-only formulations specifically labeled for pediatric use are uncommon. Families more often encounter magnesium in liquid products combined with other nutrients, such as calcium or vitamin D. These formulations require careful label review, as magnesium may not be the primary nutrient and total intake can be harder to track across multiple supplements.
As a result, liquid magnesium is best evaluated conceptually rather than assumed to be a straightforward option.
3. Chewable or Gummy Magnesium
Chewable and gummy magnesium products marketed for children are typically combination formulations, not magnesium-only supplements. They are often prioritized for taste acceptance, routine consistency, and compliance challenges. Because magnesium contributes bitterness and bulk, chewable products usually contain lower magnesium amounts alongside other nutrients or sweeteners. This makes evaluating total magnesium intake — especially when used alongside multivitamins or fortified foods — particularly important. One commonly encountered example is Olly Kids Chillax.
4. Combination Magnesium Products
Some children's supplements include magnesium incidentally as part of broader formulations, such as bone or growth-focused products that combine calcium, vitamin D, and magnesium. These products are often prioritized for convenience, but they introduce important tradeoffs: magnesium is not the primary focus, dosing flexibility is limited, and cumulative intake across supplements can be difficult to assess.
Combination products highlight why understanding context and stacking matters more than the presence of any single ingredient.
One commonly encountered example is ChildLife Essentials Liquid Calcium with Magnesium.
Examples in these categories may be referenced throughout this site. Any product mentions are selected based on formulation characteristics, not sponsorship, and are intended for educational comparison only.
Use the checklist to compare magnesium forms, dosing, and formulation tradeoffs in a more structured way.
View the Checklist →Dosing: More Is Not Better
Magnesium has established upper intake limits for supplemental forms. Excess intake — particularly from supplements — can lead to gastrointestinal upset and electrolyte imbalance.⁴
For families, this reinforces the importance of understanding magnesium from food and supplements combined, and avoiding overlap across multivitamins, gummies, and powders.⁴
Food First, Supplement Second
Many foods provide meaningful magnesium, including whole grains, legumes, nuts and seeds, and leafy greens. For children with varied diets, these sources may already contribute significantly to daily needs.
This aligns with professional guidance emphasizing food first, reserving supplementation for situations where intake, tolerance, or specific needs suggest it may be helpful.⁵
Want a more structured way to evaluate magnesium and other family supplements? The Evidence-Informed Family Supplement Guide is coming soon. It will walk through magnesium, children's multivitamins, probiotics, label claims, dosing, safety tradeoffs, and reusable worksheets — so families can make supplement decisions with more clarity and less guesswork.
See What's Coming →Why This Matters for Families
Magnesium is often added to routines with good intentions, but without clarity around form or dose. For families, thoughtful evaluation helps prevent unnecessary supplementation and reduces the risk of unintended effects — especially when products are used consistently over time.
Pulling It All Together
Magnesium decisions are rarely about identifying a single "best" form. Different forms vary in absorption characteristics, gastrointestinal tolerance, dosing flexibility, and practicality.
The most useful approach is not to search for superiority, but to clarify purpose. What is being addressed? What dose aligns with age and need? How does the chosen form fit the intended use and the child's tolerance?
In many cases, differences between forms matter less than thoughtful dosing and realistic expectations. An evidence-informed approach focuses on alignment: matching formulation, dose, and duration to context rather than reacting to marketing claims about absorption or potency.
When magnesium is considered with that lens, the decision becomes less about comparison — and more about proportional use.
Where Magnesium Fits in the Broader Supplement Conversation
If you're new to evaluating supplements, these foundational guides may help:
- How I Evaluate Supplements as a Biomedical Engineer and Quality Professional
- How to Read a Supplement Label (Without Getting Misled)
Magnesium decisions follow the same logic applied across vitamins, minerals, and probiotics: formulation quality, dose, and real-world use matter more than claims.
Browse side-by-side comparison content designed to help families evaluate supplement categories through formulation, dose, quality signals, and tradeoffs.
View All Comparisons →Get new Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families interpret supplement claims, research findings, and formulation tradeoffs more clearly.
Subscribe to the Digest →References and Further Reading
- National Institutes of Health Office of Dietary Supplements. Magnesium Fact Sheet for Health Professionals.
- Schuette SA et al. Bioavailability of magnesium diglycinate vs magnesium oxide. Journal of the American College of Nutrition.
- National Institutes of Health Office of Dietary Supplements. Magnesium Fact Sheet (Adverse Effects and Tolerance).
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes for Magnesium.
- Harvard T.H. Chan School of Public Health. Magnesium and Health.
All are publicly accessible via NIH, Harvard, or Google Scholar.
Transparency and Scope: Some links in this article may be affiliate links, which means Evidence First Wellness may earn a small commission if you choose to make a purchase — at no additional cost to you. Products are referenced for educational comparison only, and affiliate relationships do not determine editorial conclusions.
This content is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Magnesium does not need to be framed as essential for every family to be valuable. In many cases, understanding when it may not be necessary is just as important as knowing when it might be considered.
Why Dosage and Form Matter More Than Ingredient Lists
An evidence-informed explanation of why dosage and ingredient form matter more than long ingredient lists—how underdosing, bioavailability, and formulation choices affect real-world effectiveness.
A focused breakdown of how dosage and nutrient form influence effectiveness, absorption, and safety.
It is easy to give an ingredient list more weight than it deserves. It is right there on the label, and it gives you something concrete to compare: more nutrients, more compounds, more familiar names.
But more ingredients do not automatically make a formula more useful. A product can look comprehensive while still using amounts that are too low to matter, forms that are not well matched to the intended use, or a serving size that does not fit real life.
This is why dosage and form often matter more than the length of the list.
Why Ingredient Lists Can Be Misleading
Ingredient lists answer a basic question: what is included? They do not explain whether an ingredient is present in a meaningful amount, how well it is absorbed, how it behaves in the body, or whether it is appropriate for the intended user.
Two products can list the same ingredient while telling very different stories. One may provide a meaningful dose in a suitable form. The other may include an amount unlikely to matter for the stated purpose, in a form that does not perform as well. The label does not distinguish between those two scenarios.
This is why evaluating supplements requires looking beyond presence alone.
What an Ingredient List Can and Cannot Tell You
Dosage: When "Some" Isn't Enough
Most ingredients do not work simply because they are present. They are likely to matter when provided at an amount that aligns with how they have been studied or shown to function biologically.²
A supplement may include an ingredient at a dose far below what has been studied, list many ingredients at amounts that may not be meaningful for the stated purpose, or prioritize label appeal over functional relevance. In those cases, the list can look comprehensive while the amounts behind it may not be meaningful for the stated purpose.²
Dose is not about more always being better, either. The relevant question is whether the amount is appropriate for the ingredient, the intended purpose, and the person using it. Sometimes that means a lower dose is correct. Sometimes a product includes more of something than is necessary or appropriate. Context matters in both directions.
For families, this means more ingredients does not equal more benefit. Sometimes a clearer, simpler formula is the more thoughtful choice.
Presence is not the same as relevance.
An ingredient may be listed on the label, but the amount provided still needs to be meaningful enough to support the reason it was included.
Why Form Influences How an Ingredient Behaves
Ingredient form refers to the specific chemical or structural version of a nutrient or compound. Two supplements can list the same ingredient name and still deliver something meaningfully different.
The form used can affect absorption, tolerability, stability, and consistency of effect.³ A parent seeing "magnesium" or "vitamin B12" on two different labels may reasonably assume those ingredients are equivalent. Depending on the specific form used, they may not behave the same way in the body.
Labels often list the ingredient name without explaining which form was chosen or why, leaving readers to assume equivalence where there may be real differences.⁴ This is one of the most common reasons supplements do not perform as expected, even when the ingredient list looks promising.³
A one-page reference for reviewing ingredient identity, dose, form, formula quality, and supplement claims more clearly.
View Checklist →Interactions Matter More Than Lists
A supplement is not just a list of separate ingredients. It is a formula, and the ingredients have to work together in that context.
Some ingredients can enhance each other's absorption. Others can compete for uptake, affect tolerability, or change how consistently an ingredient works across different users. How ingredients behave in isolation is not always how they behave in a complete formula, particularly at lower individual doses.
This is another reason ingredient lists, by themselves, provide limited insight into how a product is likely to perform.
Before comparing two products by ingredient list alone, ask:
Practicality Is Part of Quality
For families, effectiveness is not only biological. It is also practical.
Dosage and form affect how many capsules, tablets, scoops, or gummies are required per serving. They also affect taste, texture, tolerability, and whether the product is realistic to use consistently. A product can look strong on paper and still fail in daily life. If a child will not take it, if the serving size is unrealistic, or if the format creates more friction than the product is worth, the formula may not deliver in real life.
A simpler, well-designed product that fits into daily life may be more useful than a complex one that does not.
Quality includes usability.
How This Fits Into a Thoughtful Evaluation
Looking beyond ingredient lists does not require specialized training. It requires shifting the focus from what is included to how the formula is designed to work.
A more complete evaluation considers ingredient identity, dose, form, transparency, context, and risk.
Ingredient lists are a starting point, not the conclusion.⁴
A shorter label can sometimes reflect the more thoughtful formula.
For families, the goal is not to find the longest ingredient list. The goal is to understand whether the product is clearly formulated, reasonably dosed, tolerable, and appropriate for the person who will use it.
A structured way to review evidence, dose, form, quality signals, and real-world tradeoffs before deciding whether a supplement is worth considering.
Open the Framework →Why This Matters for Families
When decisions affect children or household routines, uncertainty carries more weight. Families are not just asking whether the ingredient list looks good. They are asking whether the product is likely to help, safe and appropriate for the person using it, realistic to take, and worth adding to daily life.
Those are practical questions, and ingredient lists alone do not answer them. Understanding why dose, form, and formula design matter gives families a more useful set of things to evaluate, without requiring expertise in formulation science.
The shift is from asking "Is this ingredient included?" to asking "Is this product designed to work for the person who will use it?"
How This Connects to the Broader Framework
This focus on dose and form is part of the broader framework I use when evaluating supplements, explained in more detail in How I Evaluate Supplements as a Biomedical Engineer and Quality Professional.
That framework emphasizes how products are designed to function, not just how they are presented. That shift is what helps families move beyond surface-level comparisons and toward decisions that are more grounded in how the product is actually built.
Get new Evidence First Wellness articles and practical evaluation frameworks delivered once per week. The digest focuses on helping families interpret supplement claims, research findings, and formulation tradeoffs more clearly.
Subscribe to the Digest →References and Further Reading
- National Institutes of Health Office of Dietary Supplements. Dietary Supplement Fact Sheets, 2023.
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes (DRIs), 2019.
- Heaney RP. Factors influencing the measurement of bioavailability. American Journal of Clinical Nutrition, 2001.
- U.S. Food & Drug Administration. Dietary Supplements: What You Need to Know, 2022.
All sources are publicly accessible via NIH, FDA, or Google Scholar.
Content on this site is provided for educational purposes only and does not constitute medical advice. Individual health decisions should be made in consultation with qualified healthcare professionals.
Some links on this site may be affiliate links. Affiliate relationships do not influence editorial content, evaluation methodology, or conclusions. For more information, see the Disclosure & Affiliate Policy.